|
|
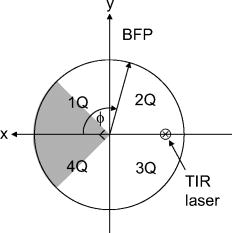
1.IntroductionFluorescent markers are highly sensitive, potentially single-molecule, detectors used in cells, tissues, and other biological specimens. They can identify and characterize low abundance substrates in the visible wavelengths where biological specimens have low autofluorescence and where marker fluorescence is efficiently collected in a microscope objective. They are also direction sensors due to the electric dipole–mediated excitation and fluorescence-coupled decay. The electric dipole is nominally fixed in the molecular frame of the fluorophore that is likewise fixed relative to the substrate. Absorption and fluorescence emission from the dipole is recognized to obey absorption probability and emitted intensity angular distribution rules leading to deduction of probe orientation from collected polarized fluorescence intensities. The fluorescence microscope is an imaging instrument producing a real image of the fluorescence source at a light detector. Nevertheless, accurate spatial representation of the fluorescent source might be secondary to efficient light detection in some applications although resolution and light detection efficiency tend to improve together with increasing numerical aperture (NA) of the microscope objective. A single-molecule fluorescence polarization application in which the sources are sufficiently separated or selectively activated only needs microscope collection efficiency.1, 2 In this case, an image of the objective back focal plane (BFP) light intensity distribution contains all the dipole orientation information in a pattern that is interpretable by inspection.3, 4 The fluorescence microscope is an efficient emitted light-collecting instrument that can also perturb the emission intensity angular distribution that we wish to measure. The perturbation is complex in a microscope utilizing total internal reflection excitation (TIR). In TIR microscopy, excitation light impinges on a glass/aqueous interface from the glass side at angles beyond critical angle for TIR. The light totally reflects and remains inside the glass substrate except for an evanescent layer of excitation energy in the aqueous phase that decays exponentially in the direction normal to the interface. The evanescent excitation excites fluorescence from the sample in the aqueous phase in a region within of the interface. At this short distance, the interface alters the emission angular pattern and total emitted energy of the fluorophore as described by Hellen and Axelrod.5 The interface converts emitter dipole nonpropagating (near-field) into propagating (far-field) energy density collected by the high-NA objective. Additionally, the high-NA collecting lens intermingles emitter linear polarization components again predictably perturbing the emission angular pattern.6 To varying degrees, both effects enrich the information content of the BFP pattern and complicate its quantitative interpretation. Here we describe efforts to calculate and observe the two-dimensional BFP emission pattern for a high-NA microscope objective. Patterns were observed under TIR illumination of bare glass/aqueous and glass/thin-aluminum-film/aqueous interfaces. Samples observed included fluorescent nanospheres and single photoactivatable green fluorescent proteins (PAGFP) tagged myosin light chains exchanged onto myosin cross-bridges in skeletal muscle fibers. The latter is an interesting biological application on crowded proteins, where emission from a single brightly fluorescent probe competes with background from many more probes in the low-fluorescence state.2 Our results show good agreement between observed and calculated patterns for the fluorescent nanosphere sample where sample intensity dominates the pattern. In contrast, while single photoactivated PAGFPs in the muscle fiber sample create a distinctive image even while embedded in background from unphotoactivated PAGFPs, the BFP pattern for the photoactivated PAGFP is much less distinctive. By collecting the BFP pattern at a nontelecentric plane, we can partially separate the single-molecule BFP pattern from the background BFP pattern. The latter method made possible a qualitative interpretation of probe dipole orientation and has potential for further development. 2.Methods2.1.ChemicalsCarboxylate-modified fluorescent microspheres were purchased from Molecular Probes (Eugene, Oregon). Adenosine triphosphate (ATP), dithiothreitol (DTT), and phenylmethanesulfonyl fluoride (PMSF) were from Sigma Chemical (St. Louis, Missouri). All chemicals were analytical grade or ultrapure, if available. 2.2.SolutionsRigor solution contains imidazole, ethylene glycol bis( -aminoethyl ether)-N,N, -tetraacetic acid, Mg acetate, potassium propionate, PMSF, leupeptin, and DTT. 2.3.Sample PreparationsRabbit psoas muscle fibers were prepared, and the native myosin regulatory light chain (RLC) exchanged with PAGFP-tagged human cardiac RLC (HCRLC-PAGFP) as described.1, 2 Red-orange carboxylate-modified fluorescent spheres had diam and excitation/emission maxima at . Sphere concentrations were computed using the formula from the manufacturer and diluted in rigor buffer. We used -fold dilutions from stock giving sphere concentration of . All experiments were conducted at room temperature. 2.4.Coverslip PreparationClean No. 1 glass coverslips were sonicated for in ethanol then plasma cleaned (Harrick Plasma, Ithaca, New York) for . Plasma-cleaned coverslips were ready for use in the bare glass interface experiments. Plasma-cleaned coverslips were also thin aluminum (Al) film coated by thermal vacuum evaporation at the Mayo Electron Microscope Facility. In some instances, film deposition was limited to half of the coverslip’s top surface by partially covering one coverslip with another during vacuum evaporation. The result was an abrupt edge to the film as viewed in the light microscope. Aluminum film depth was measured with atomic force microscopy by scanning the tip over the edge in the partially aluminized coverslip. Aluminum film depth was estimated to be . 2.5.Sample Chamber PreparationBare glass or Al-coated coverslips were placed on a brass slide with a large hole cut out, permitting the objective from the inverted microscope to contact the coverslip through immersion oil. The Al-coated coverslips had the metal side up. A watertight chamber was constructed on top of the coverslip as described.7 The chamber contained either a suspension of nanospheres or a muscle fiber. Nanospheres settled from suspension onto the coverslip, where some of them adsorbed strongly enough to remain after washing out free nanospheres with rigor buffer. Adsorbed spheres were often aggregates with varying emitted intensities. BFP images were from microsphere aggregates. 2.6.TIR Fluorescence BFP MicroscopyThe inverted microscope (Olympus IX71) setup was described previously.1 Excitation 488 or light from an argon-ion laser is focused on the BFP of a 1.45-NA objective and incident from the glass side of a glass/aqueous interface at angles greater than critical angle for TIR, (see Fig. 1 ). Although light is totally reflected, an evanescent field created in the water medium and decaying exponentially with distance from the interface, excites fluorophores within of the surface.8 The p-polarized incident light has electric field polarization in the incidence plane and produces an elliptically polarized evanescent electric field. Evanescent intensity is predominantly polarized normal to the interface.9 The s-polarized incident light has electric field polarization perpendicular to the incidence plane that is continuous across the interface. In the presence of the metal film, the laser penetrates the interface and creates an evanescent field only when incident at the surface plasmon resonance angle, (see Fig. 1). P-polarized incident light penetrates the interface while s-polarized light is mostly reflected. Experiments with green fluorescent protein (GFP)-tagged muscle fibers use reduced illumination area on the fiber surface accomplished with the laser diameter reducing aperture shown in Fig. 1. Fig. 1Lens L focuses 488 or of light from an argon-ion laser at the BFP of a 1.45 NA oil-immersion objective. The light is incident from the glass side of the glass/aqueous or glass/metal-film/aqueous interface at angles greater than critical angle, , or equal to the surface plasmon resonance angle, . The excitation aperture reduces the illuminated area in the sample plane. 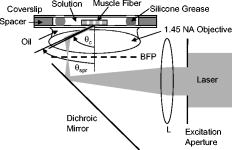 Fluorescent spheres or HCRLC-PAGFP exchanged muscle fibers in aqueous buffer solution make contact with the TIR-supporting glass or metal-film substrate and are illuminated by the evanescent field. Excited fluorescence is collected by the objective and spectrally filtered using a rhodamine (fluorescent spheres) or fluorescein (GFP-tagged fibers) filter set. Figure 2 shows the optical setup. Fig. 2Optical setup for conventional and BFP pattern imaging. The object (black arrow) is at the object plane (OP), OBJ is objective, SP is the plane where the BFP pattern (double arrow) is observed, TL is tube lens, RL is a removable lens, CCD is the image plane for the camera, and is the tube lens focal length. Light emitted by the object at OP emerges from the back of the objective in parallel rays. RL is removable (bidirectional arrow) to facilitate BFP (RL in) or object (RL out) imaging. 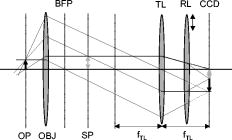 The BFP lies within the Olympus objective and is not readily accessible, hence, we image the intensity pattern at the BFP from a sample plane (SP) between the BFP and tube lens (TL). Imaging an object at SP (light double arrow) rather than the BFP changes the image size at the CCD plane. SP is not telecentric, implying an object displaced laterally changes the position of its intensity pattern at SP (the pattern at a telecentric plane is not translated when the object moves laterally). All experiments were conducted with the object at the center of the field. A single molecule from a muscle fiber was centered in the field, but most of the background fluorescence was intentionally displaced from the center likewise displacing its BFP pattern at SP. The background BFP pattern displacement enhances our ability to separately identify BFP patterns for the single molecule and background. The removable lens (RL) is positioned to form a real image of SP at the CCD plane by placing a translucent ruler at SP and adjusting RL position until a real image of it is formed at the CCD plane. The RL is conveniently removable (indicated in Fig. 2 by the two-directional arrow) to facilitate conversion between BFP and normal imaging. 2.7.Emission Near an InterfaceWhen a dipolar is near a conducting or dielectric planar interface, the radiated fields are significantly altered.5 An emitting fluorophore has a dipolar emission field that is the superposition of propagating transverse waves forming the far-field and nonpropagating longitudinal (evanescent) waves forming the near field. The near field is not detected unless perturbed by a nearby interface creating detectable propagating transverse waves. Converted near-field propagating waves appear in the glass medium at angles beyond for the dielectric interface or at for the metal-coated interface. We refer to either emission mode as supercritical angle fluorescence (SAF).10 Oil immersion objectives with capture SAF. Subcritical angle fluorescence (UAF) is from far-field emission. Figure 3 shows emission near a bare glass (left) or metal-film (right) interface and indicates the critical angle, , and surface plasmon resonance (SPR) angle, . The objective lens subtends a polar angle appropriate for a objective. Fig. 3Far- and near-field emission from dipole transmits the interface and propagates into the glass medium at angles beyond critical angle, , for the dielectric interface or at the SPR angle, , for the metal-coated interface. SAF is supercritical angle fluorescence referring to any near-field light that propagates into the region defined by . The objective lens drawn subtends a solid angle appropriate for a objective. The BFP and laser beam for TIR excitation are also shown. 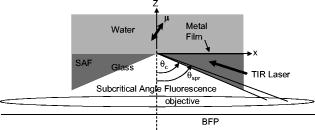 The interface also affects total radiated power. Hellen and Axelrod pointed out that, for a fluorophore under steady illumination, the dissipated power must equal the absorbed power, implying that a fixed power, rather than a fixed amplitude, dipole radiator is the appropriate model for probe emission near an interface.5 An important consequence of the power normalization is that the metal film totally quenches fluorescence from probes within of the interface. Hellen and Axelrod also pointed out conditions where reflection coefficients for the conducting interface need to incorporate nonlocal effects due to the dipole’s near-field coupling to surface waves whose wavelengths do not satisfy the condition of being much greater that the mean free path of conducting electrons in the metal. They use reflection coefficients derived from quasistatic semiclassical infinite barrier (SCIB) theory.11 The SCIB correction, inserted when large wavenumber radial waves of the dipole’s near-field contribute to energy dissipation, is largest for dipoles very near to the metal film and affects total power dissipated by the dipole. The high-NA collection of light in a microscope objective mixes light contributions from emitter dipole Cartesian components due to the collection of light from a large solid angle surrounding the dipole. The Cartesian dipole moments produce differently polarized light that is combined by the objective such that their individual contributions cannot be separated using an analyzing polarizer. Separating the contributions into the Cartesian components of the dipole moment is the basis for fluorescence polarization measurement of dipole orientation. Axelrod described the solution to the problem in a homogeneous medium.6 We address the probe emission perturbation by the interface and the complications for polarized light collection by a high NA objective in the dipolar emission intensity from single probes. Our computations give the two-dimensional intensity pattern in the objective BFP for comparison to experiment and show the effect of probe orientation on the pattern. Computation and display of BFP intensity patterns were performed using software written for Mathematica (version 6.0, Wolfram Research, Champaign, Illinois). 3.Results3.1.Two-Dimensional BFP IntensityA dipole in aqueous medium near to a bare glass or metal-film-coated glass interface emits light part of which transmits to the glass side and is collected by the objective. Figure 4 shows the spatial relationship of the dipole moment, interface, objective, BFP, SP, pattern mask (MA), filter set (FS), and analyzing polarizer (AP). Fig. 4The spatial relationship of the dipole moment , interface ( plane), objective, BFP, sample plane (SP), pattern mask (MA), filter set (FS), and analyzing polarizer (AP). The bare glass or metal-film-coated glass side of the interface lies in the half-plane and the aqueous side in the half-plane. The - and -auxiliary lines lie in the plane. Dipole is at the focal point of the objective with -axis position and orientation in the microscope fixed frame given by . Heavy arrows delineate an emitted light ray path. The ray is incident on the objective with propagation vector and emerges in image space at distance from the -axis, where is the objective focal length and is the refractive index for the objective and immersion oil. Coordinates define the observation point at the BFP or SP. 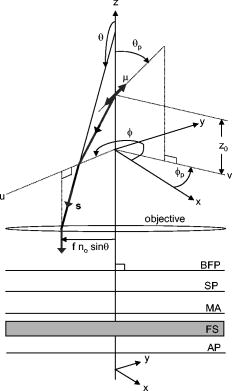 Heavy arrows delineate an emitted light ray path. It leaves the dipole, refracts at the interface and at the objective, then emerges in image space propagating parallel to the -axis. The ray is incident on the objective with propagation vector and emerges in image space at distance from the -axis, where is the objective focal length and is the objective and immersion-oil refractive index. Coordinates define the observation point at the BFP. We computed the intensity at SP using the method outlined previously1 and based on the original work of Hellen and Axelrod.5 A fluorescent object translated laterally from the center of the visual field produces the BFP pattern like the centered object implying multiple single molecules in the field produce a summed BFP pattern. The pattern image at SP however is translated with object translation. The translated image is approximated using a coordinate transformation that accounts for the changed light collection geometry but not for the changed light refraction at the objective. The latter would be needed to interpret the translated BFP pattern quantitatively. An object translated a lateral distance from at the visual field center is one focus of an ellipse with the other focus at the visual field center. The semimajor axis and eccentricity for the ellipse are Electric fields computed for the object in coordinates were expressed in coordinates fixed in the visual field center using the following relationships:for the ellipse radius measured from the visual field center focus.Computed BFP intensity patterns are averaged over square regions to imitate pixilation by a CCD camera. Pixels correspond to regions in image space as appropriate for our CCD camera (Hamamatsu, Orca ER). Figure 5 compares computed BFP intensity patterns for a bare glass interface with a dipole at and orientation , and (top) or (bottom). Patterns for dipole orientation have a reflection symmetry axis along a line at ; hence, we can compare patterns with a half-plane representation. All patterns show an intense circular band where critical angle light intersects the BFP (arrows). Light falling outside the band is SAF, and light falling within the band is UAF. Figure 5a shows the intensity pattern without the AP. The two dipoles and are subtly distinguished in the pattern mainly with UAF intensity near the equatorial (horizontal line bisecting the circular pattern). Figures 5b and 5c show the BFP patterns after analyzing polarizer (AP) insertion for active axis at two perpendicular directions indicated by the white arrows in the right bottom corners. The patterns distinguish the or dipoles in both SAF and UAF intensity but SAF is the more characteristic indicator of dipole orientation. Fig. 5Computed BFP intensity patterns for a bare glass interface for dipoles at and orientation , and (top), or (bottom) with -axis indicated and -axis pointing into the page. All patterns show an intense circular band where critical angle light intersects the BFP (arrows inside ring). Light falling outside the band is SAF, and light falling within the band is UAF. (A) shows the intensity pattern without the AP. Dipoles with and components (and with ) are distinguished in the pattern mainly with UAF intensity near the equatorial (horizontal line bisecting the circular pattern). (B, C) show the BFP patterns after analyzing polarizer (AP) insertion for active axis at two perpendicular directions indicated by the white arrows in the right bottom corners. The patterns distinguish dipoles equivalent except for the and components in both SAF and UAF intensity. BFP patterns in (A) and (B) differ at 10:30 and 4:30 o’clock where (A) is brighter in the UAF and SAF regions.  The panels demonstrate that an important degeneracy in dipole moment orientation is lifted by BFP imaging. In conventional microscope fluorescence polarization, the dipole orientations , , and will produce equal intensities. The high-NA objective collects Cartesian dipole component products , , and in addition to the usual products and collected at low NA. The BFP pattern shows the functional dependence on . The combination (high NA and dependence in the BFP pattern) removes the , degeneracy, but , degeneracy remains. Conventional object imaging microscopy sums intensity uniformly over reducing to zero weights for the , , and products thereby removing their contribution from detected intensity. Figure 4 shows the position of the mask within the microscope, which can be configured to nonuniformly sample contributions from different ’s reinstating , , and sensitivity in the total detected intensity. Figure 6 shows angle and BFP subdivision into four quadrants. Emitted light propagates toward the viewer and the excitation TIR laser beam propagates into the page. Emitted light is blocked from propagating beyond the BFP in the shaded region. The presence of the mask removes the , degeneracy. With the mask from Fig. 6, collected intensities for the system depicted in Fig. 5a with and have relative values of 1 and 0.6, respectively. Under the same circumstances, the intensity ratio for the systems depicted in Figs. 5b and 5c [the intensity collected in Fig. 5b divided by the intensity collected in Fig. 5c] is 1 and 2.3 for and , respectively. 3.2.Quantitative BFP Pattern ModelingSmall aggregates of fluorescent nanospheres were adsorbed onto Al-coated or bare glass coverslips then illuminated under TIR. Figure 7a compares experimental (top half) and computed (bottom half) BFP patterns for the nanospheres near the Al-coated interface. The inner ring appears at (inner arrow, bottom left) position and the outer ring at the position (outer arrow bottom left). Fig. 7(A) contains experimental (top half) and computed (bottom half) BFP patterns for nanospheres near the Al-coated interface. The inner ring appears at the critical angle, , position (inner arrow) and the outer ring at the surface plasmon resonance angle, , position (outer arrow). (B) compares equivalent projections of the experimental and computed patterns. The computed pattern uses the parameters indicated in (B). 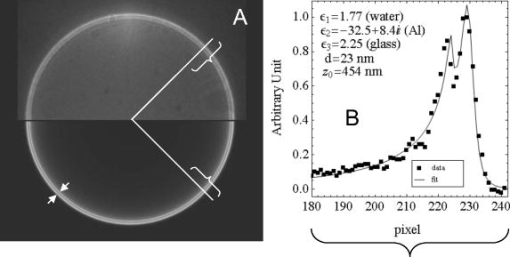 Figure 7b compares experimental and computed patterns showing good alignment of peaks. The parameters needed in the fitting are the dielectric constants for water, glass, and aluminum; the thickness of the metal film ; the distance of the dipole from the interface ; and the orientation of the dipole . The dipole orientation was assumed to be perpendicular to the interface due to the apparent cylindrical symmetry of the rings. We did not investigate other polar angles with cylindrical symmetry imposed by averaging over . Dielectric constants were taken from Ref. 5 while parameters and were varied to find a suitable fit. We find that fitted falls within the experimentally determined Al-film depth of . Fitted dipole position is beyond the expected penetration depth of the TIR evanescent field of . The discrepancy suggests that the nanosphere aggregate frustrates TIR of the incident beam to propagate excitation into the aggregate. Figure 8 compares experimental (top half) and computed (bottom half) BFP patterns for the nanospheres aggregate near a bare glass interface. The broad bright ring begins at the critical angle position but continues into the SAF region, indicating probes are emitting very near to the interface. The faint ring patterns seen in the UAF region not reproduced in simulation are probably caused by diffraction of scattered excitation laser light contaminating the image from fluorescence. The same uneven background effect probably produces the faint dark band in the outermost SAF region also not reproduced in simulation. The experimental BFP emission pattern is heterogeneous in and slightly more intense at the meridian (vertical line bisecting the circular pattern) than at the equatorial line and qualitatively similar to the computed pattern where and . Fig. 8Experimental (top half) and computed (bottom half) BFP patterns for the nanospheres aggregates near a bare glass interface. The broad bright ring starts at the critical angle, , and continues into the SAF region indicating probes are emitting very near to the interface. The computed pattern uses and .  Figure 9a shows a HCRLC-PAGFP exchanged muscle fiber in rigor at a bare glass interface and under TIR illumination. Emission is dominated by a single photoactivated PAGFP. Background light is from many unphotoactivated HCRLC-PAGFPs in the fiber emitting at the efficiency of the photoactivated molecule with excitation. The exciting TIR laser aperture is closed down (Fig. 1) to minimize the area illuminated around the photoactivated molecule. Further reduction in aperture size unacceptably reduced fluorescence intensity from the photoactivated single molecule. The single dominant photoactivated HCRLC-PAGFP was positioned at the center of the microscope field of view and at the lower edge of the illuminated region such that the majority of background emission originated from the top portion of the field of view. Fig. 9(A) A HCRLC-PAGFP exchanged muscle fiber in rigor at a bare glass interface and under TIR illumination. The single dominant photoactivated HCRLC-PAGFP is at the center of the microscope field of view and at the lower edge of the illuminated region. (B) The BFP image from (A) and simulated BFP image from an object at the center plus an object translated upward from center of the field of view (right and left hemicircles, respectively). The single dominant photoactivated HCRLC-PAGFP emission in the fiber image maps to the centered ring at the outer edge of the BFP pattern. Background in the fiber image maps to the downward translated broad BFP ring pattern. 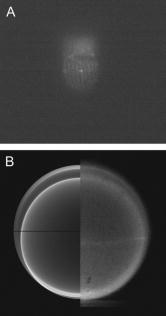 The right side of Fig. 9b shows the BFP image from Fig. 9a. The left side of Fig. 9b shows the simulated BFP image from two objects, one at the center and another translated upward from center of the field of view. An object translated laterally produces a laterally translated BFP image at SP. The object translated upward produces the simulated light ring translated downward in correspondence with the broader observed light ring at the BFP. In the fiber image, background is centered upward from the single molecule source but is distributed over the whole illuminated region producing the translated and diffuse light ring in the BFP image. Simulating the distributed background sources over the whole illuminated region is prohibitively computer time consuming and was not attempted. The object at the center of the field produces the centered simulated light ring in correspondence with the centered observed light ring in the BFP image. The translated background BFP image allows observation of the centered object over about half of the BFP image. The single-molecule source BFP image diminishes at the equator, indicating an oriented single molecule. The simulated ring is identical to that in the lower part of Fig. 5a, where and . 4.DiscussionSingle fluorescent probe dipole orientation is recognized by its two-dimensional emission pattern at the BFP of a high-NA microscope objective.3 For the system depicted in Fig. 3 with dipole excited by TIR light, the BFP fluorescence emission pattern has intensity concentrated at the critical angle for the bare glass interface (Fig. 5) and at the critical angle and surface plasmon resonance angle for the thin-metal-film–coated glass interface [Fig. 7a]. These distinctive patterns show that a substantial portion of light transmitted into the glass side of the interface is SAF for the bare glass interface and essentially all transmitted emission is SAF and collected into a cone at in the presence of the thin metal film. Selecting the objective NA large enough to collect this light is essential for maximizing emitted signal collection efficiency.4 The depends on the dielectric constant of the metal film, providing an adjustable parameter to facilitate collection of emitted light. As pointed out by Mattheyses and Axelrod, the for an Al film is suited for use with the TIRF objective.4 The BFP pattern from isolated and immobilized single probes determined emitter dipole orientation with good accuracy3 when compared to alternative emission methods such as defocused orientation imaging12, 13 and other polarization analysis procedures.12 The BFP patterns distinguish in principal all dipole orientation changes aside from the degeneracy when the dipole is inverted through the origin. The BFP intensity pattern thus contains dipole orientation information missing from conventional spot intensity fluorescence polarization microscopy. Figure 5a, 5b, 5c demonstrate the effect by indicating the ability to distinguish and dipole orientations that are degenerate in conventional microscopy. The degeneracy is lifted because Cartesian dipole component cross products , , and , (where , , and ) are detected (in addition to the usual products and ) with the high-NA objective and the BFP intensity directly shows their effect on the emission pattern. In conventional fluorescence microscopy, the cross products are detected, but this information is lost when the collected intensity is uniformly summed over . Without BFP pattern information, lost information is recoverable using the mask shown in Figs. 4 and 6. The mask nonuniformly weights contributions from different ’s recovering , , and sensitivity in the detected intensity. In muscle, myosin is the actin-dependent molecular motor driving sarcomeric shortening and muscle contraction by transducing chemical free energy from MgATP hydrolysis into directed movement.14, 15 The MgATP hydrolysis is coupled to a series of conformational changes in myosin resulting in the myosin lever arm rotation to impel actin against resistive force.15 A GFP-tagged myosin light chain (MLC-GFP) was exchanged with the native light chain on the lever arm to fluorescent label the myosin lever arm in a muscle fiber. The MLC-GFP rigidly attaches to the lever arm to detect its rotation with fluorescence polarization.1 In the fiber, myosin concentration reaches ,16, 17 implying myosin is crowded and complicating isolation of individual molecules for single-molecule measurements. Recently, photoactivatable GFP-tagged MLC (MLC-PAGFP) was introduced to the fiber.2 Sparse photoactivation of the PAGFP permitted detection of single-lever-arm orientation in the myosin dense muscle fiber. A persistent difficulty in interpreting fluorescence from the single photoactivated MLC-PAGFP–tagged myosin is background light from many unphotoactivated MLC-PAGFPs in the fiber emitting at the efficiency of the photoactivated molecule. Figure 9a shows a single photoactivated MLC-PAGFP on myosin in the muscle fiber. Laser TIR illumination is nominally uniform in the lateral dimension of object space out to the edge formed by the illumination aperture (Fig. 1), where it drops off rapidly. We reduced background light reaching the BFP by minimizing the overall illuminated region and spatially isolated most of the background light in the BFP pattern by placing the single photoactivated molecule at the center of the viewing field but at the edge of the illuminated region. This arrangement led to the BFP pattern shown in Fig. 9b, where the single-molecule pattern is translated from much of the background light BFP pattern. Total background light is larger than the single-molecule light; nonetheless, about half of the single-molecule BFP pattern is recognized. More background reduction is needed to make the approach quantitative. Introduction of an Al-film at the interface is one proven background reducing approach we are pursuing.18 Alternatively, the single-molecule image of Fig. 9a separates background from signal. The single-molecule image analyzed by standard spot intensity fluorescence polarization, whereas using the emission mask from Fig. 6 will characterize dipole orientation at the resolution level of the BFP pattern albeit without the convenience of recognizing orientation by inspection. Dipolar fluorescence emission characteristics in homogeneous space are variously affected when the dipole emits near an interface. The effects are caused by interference of incident and reflected emission on the dipole side of the interface, by conversion of the dipole near field into propagating light across the interface, and by normalization of the dipole emission with the total dissipated power. Fluorescence lifetime, radiated power,5, 19, 20 and the radiated power angular distribution are all affected by the interface.5, 21, 22 The power-normalized dipole emission gave the corrected radiated power angular distribution5 and lead to the dramatic observation of metal-film quenching of chromophores within a few nanometers of the metal surface.18, 23 The effect of near field on power-normalized emission transmitted into the denser medium for the dielectric (bare) interface, especially when a metal film coats the dielectric, has been transformative in spectroscopy and microscopy. Surface plasmon-coupled emission (SPCE) occurs when a fluorophore is near a metal-coated dielectric interface and causes emission, transmitting the interface to be uniquely polarized and condensed into a narrow cone of light.24 TIR illumination of the metal-coated interface produces evanescent excitation light due to the SPR effect in the metal.25 SPR excitation and SPCE together minimize the detection volume and enhance emitted light collection efficiency, both critical in single-molecule isolation and detection.18, 26 BFP pattern imaging in a microscope gives the opportunity to distinguish the various emission modes (UAF, SAF/SPCE), because all are spatially separated in the pattern. The BFP pattern imaging is also consistent with all excitation modes, including TIR and SPR. 5.ConclusionsWe have shown that, through the objective, TIR excitation and BFP pattern imaging give interpretable results related to fluorescent dipole orientation and distance from the interface. Theoretical consideration of this system has led to identification of the specific emission characteristics that remove a significant dipole orientation degeneracy found in standard microscope fluorescence polarization measurements. The degeneracy is lifted by inspection of the BFP pattern but can also be removed in a standard microscope by the introduction of a BFP mask that partially blocks some of the emitted light. The BFP pattern from single photoactivated PAGFP-tagged myosin in a muscle fiber was observed despite the large background light from the highly concentrated myosin tagged with unphotoactivated PAGFP. This was accomplished by moving the pattern imaging plane to a nontelecentric plane, where the bulk of the background intensity is translated at a distance related nonlinearly to its lateral distance from the single-molecule object. ReferencesT. P. Burghardt, K. Ajtai, D. K. Chan, M. F. Halstead, J. Li, and Y. Zheng,
“GFP tagged regulatory light chain monitors single myosin lever-arm orientation in a muscle fiber,”
Biophys. J., 93 2226
–2239
(2007). https://doi.org/10.1529/biophysj.107.107433 0006-3495 Google Scholar
T. P. Burghardt, J. Li, and K. Ajtai,
“Single myosin lever-arm orientation in a muscle fiber detected with photoactivatable GFP,”
Biochemistry, 48 754
–765
(2009). https://doi.org/10.1021/bi8017703 0006-2960 Google Scholar
M. A. Lieb, J. M. Zavislan, and L. Novotny,
“Single-molecule orientations determined by direct emission pattern imaging,”
J. Opt. Soc. Am. B, 21 1210
–1215
(2004). https://doi.org/10.1364/JOSAB.21.001210 0740-3224 Google Scholar
A. Mattheyses and D. Axelrod,
“Fluorescence emission patterns near glass and metal-coated surfaces investigated with back focal plane imaging,”
J. Biomed. Opt., 10 054007
(2005). https://doi.org/10.1117/1.2052867 1083-3668 Google Scholar
E. H. Hellen and D. Axelrod,
“Fluorescence emission at dielectric and metal-film interfaces,”
J. Opt. Soc. Am. B, 4 337
–350
(1987). https://doi.org/10.1364/JOSAB.4.000337 0740-3224 Google Scholar
D. Axelrod,
“Carbocyanine dye orientation in red cell membrane studied by microscopic fluorescence polarization,”
Biophys. J., 26 557
–573
(1979). https://doi.org/10.1016/S0006-3495(79)85271-6 0006-3495 Google Scholar
T. P. Burghardt, K. Ajtai, and J. Borejdo,
“In situ single molecule imaging with attoliter detection using objective total internal reflection confocal microscopy,”
Biochemistry, 45 4058
–4068
(2006). https://doi.org/10.1021/bi052097d 0006-2960 Google Scholar
D. Axelrod and G. M. Omann,
“Combinatorial microscopy,”
Nat. Rev. Mol. Cell Biol., 7 944
–952
(2006). https://doi.org/10.1038/nrm2062 1471-0072 Google Scholar
T. P. Burghardt and N. L. Thompson,
“Evanescent intensity of a focused gaussian light beam undergoing total internal reflection in a prism,”
Opt. Eng., 23 62
–67
(1984). 0091-3286 Google Scholar
T. Ruckstuhl and D. Verdes,
“Supercritical angle fluorescence (SAF) microscopy,”
Opt. Express, 12 4246
–4254
(2004). https://doi.org/10.1364/OPEX.12.004246 1094-4087 Google Scholar
G. W. Ford and W. H. Weber,
“Electromagnetic interactions of molecules with metal surfaces,”
Phys. Rep., 113 195
–287
(1984). https://doi.org/10.1016/0370-1573(84)90098-X 0370-1573 Google Scholar
E. Toprak, J. Enderlein, S. Syed, S. A. McKinney, R. G. Petschek, T. Ha, Y. E. Goldman, and P. R. Selvin,
“Defocused orientation and position imaging (DOPI) of myosin V,”
Proc. Natl. Acad. Sci. U.S.A., 103 6495
–6499
(2006). https://doi.org/10.1073/pnas.0507134103 0027-8424 Google Scholar
A. P. Bartko and R. M. Dickson,
“Three-dimensional orientations of polymer-bound single molecules,”
J. Phys. Chem. B, 103 3053
–3056
(1999). https://doi.org/10.1021/jp9846330 1089-5647 Google Scholar
H. E. Huxley,
“The mechanism of muscular contraction,”
Science, 164 1356
–1366
(1969). https://doi.org/10.1126/science.164.3886.1356 0036-8075 Google Scholar
H. Onishi and M. F. Morales,
“A closer look at energy transduction in muscle,”
Proc. Natl. Acad. Sci. U.S.A., 104 12714
–12719
(2007). https://doi.org/10.1073/pnas.0705525104 0027-8424 Google Scholar
C. R. Bagshaw, Outline Studies of Biology: Muscle Contraction, Chapman and Hall, London
(1982). Google Scholar
B. A. Tikunov, H. L. Sweeney, and L. C. Rome,
“Quantitative electrophoretic analysis of myosin heavy chains in single muscle fibers,”
J. Appl. Physiol., 90 1927
–1935
(2001). 8750-7587 Google Scholar
T. P. Burghardt, J. E. Charlesworth, M. F. Halstead, J. E. Tarara, and K. Ajtai,
“In situ fluorescent protein imaging with metal film-enhanced total internal reflection microscopy,”
Biophys. J., 90 4662
–4671
(2006). https://doi.org/10.1529/biophysj.105.079442 0006-3495 Google Scholar
W. Lukosz and R. E. Kunz,
“Light emission by magnetic and electric dipoles close to a plane dielectric interface. I. total radiated power,”
J. Opt. Soc. Am., 67 1607
–1614
(1978). https://doi.org/10.1364/JOSA.67.001607 0030-3941 Google Scholar
K. H. Drexhage,
“Interaction of light with monomolecular dye layers,”
Progress in Optics, 163
–232 North-Holland Publishing, Amsterdam
(1974). Google Scholar
W. Lukosz and R. E. Kunz,
“Light emission by magnetic and electric dipoles close to a plane dielectric interface. II. radiation patterns of perpendicular oriented dipoles,”
J. Opt. Soc. Am., 67 1615
–1619
(1978). https://doi.org/10.1364/JOSA.67.001615 0030-3941 Google Scholar
T. P. Burghardt and N. L. Thompson,
“Effect of planar dielectric interfaces on fluorescence emission and detection: evanescent emission with high aperture collection,”
Biophys. J., 46 729
–738
(1984). https://doi.org/10.1016/S0006-3495(84)84071-0 0006-3495 Google Scholar
R. M. Fulbright and D. Axelrod,
“Dynamics of nonspecific adsorption of insulin of etythrocyte membranes,”
J. Fluoresc., 3 1
–16
(1993). https://doi.org/10.1007/BF00865284 1053-0509 Google Scholar
I. Gryczynski, J. Malicka, Z. Gryczynski, and J. R. Lakowicz,
“Radiative decay engineering 4. experimental studies of surface plasmon-coupled directional emission,”
Anal. Biochem., 324 170
–182
(2004). https://doi.org/10.1016/j.ab.2003.09.036 0003-2697 Google Scholar
D. Axelrod,
“Total internal reflection fluorescence microscopy in cell biology,”
Methods Enzymol., 361 1
–33
(2003). https://doi.org/10.1016/S0076-6879(03)61003-7 0076-6879 Google Scholar
J. Borejdo, Z. Gryczynski, N. Calander, P. Muthu, and I. Gryczynski,
“Application of surface plasmon coupled emission to study of muscle,”
Biophys. J., 91 2626
–2635
(2006). https://doi.org/10.1529/biophysj.106.088369 0006-3495 Google Scholar
|