|
|
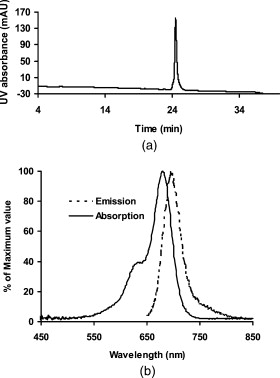
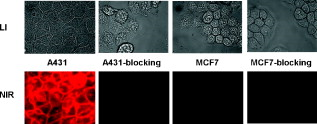
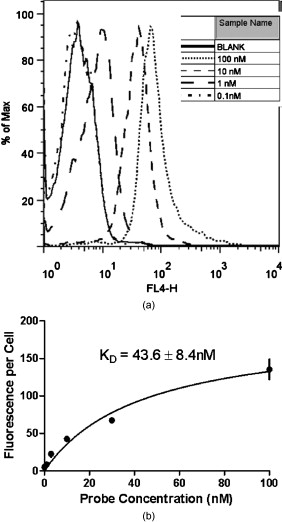
1.IntroductionEpidermal growth factor receptor (EGFR) is a transmembrane protein belonging to ErbB oncogene family. Overexpression of EGFR has been frequently detected in a wide range of human tumors, for example, non-small-cell lung cancer, small-cell carcinoma of the head and neck, esophageal cancer, gastric cancer, gliomas, colon cancer, pancreas cancer, breast cancer, ovarian cancer, bladder cancer, kidney cancer, and prostate cancer.1 Recently, EGFR has emerged as an attractive target for cancer therapy. EGFR-targeted therapies including the use of monoclonal antibodies (MAbs: cetuximab, panitumumab) and tyrosine kinase inhibitors [TKIs: gefitinib (Iressa), erlotinib (Tarceva), lapatinib (Tykerb)] have been developed and extensively studied.2, 3, 4 Meanwhile, molecular probes for EGFR imaging have also been under active investigation.5, 6, 7 It is expected that EGFR-specific molecular probes could potentially be used for early detection of EGFR positive tumors and metastases, prediction of efficacy for EGFR-targeted therapy, and for guidance in surgery etc. A variety of small-molecule-based EGFR ligands and anti-EGFR antibodies are available for developing molecular probes for different imaging modalities. However, it has been found that small molecules labeled with radionuclides generally showed rapid blood clearance, but very low tumor uptake and poor tumor imaging quality.5, 6, 7 Moreover, these small molecules are relatively sensitive to chemical modifications with large bulky tags, and they may not be a good platform for developing optical imaging probes for EGFR. On the other hand, fluorescent dyes or radiolabeled anti-EGFR MAbs have been prepared and have demonstrated good tumor uptake. However, because of slow tumor targeting ability and slow clearance8, 9, 10 of the MAbs, the tumors can be visualized only at many hours or even days after injection of the probes. Substantial improvement of EGFR molecular probes is still required for preclinical and clinical applications. Small protein scaffolds have shown great potential for recognizing a variety of biomarkers.11, 12 Among them Affibody molecules are a promising generalizable platform for developing imaging or therapeutic agents for different molecular targets.13, 14, 15 Affibody molecules are small, engineered proteins with only 58 amino acid residues and a three-helix bundle scaffold structure, while they display a binding surface as large as antibodies. High-affinity Affibody proteins (lower nanomolar or even picomolar) against different targets could be obtained through the phage display technique and affinity maturation. Because of the small size and high affinities, Affibody proteins generally show fast tumor targeting (within a half hour), high tumor uptake, and quick clearance from normal tissues. Moreover, Affibody proteins can be chemically synthesized with reasonable yield using conventional solid-phase peptide synthesis. Synthetic Affibodies retain the high binding specificity and affinity. Anti-human epidermal growth factor receptor 2 (HER2) Affibody molecules have been labeled with various radionuclides, organic dyes, and nanoparticles for imaging applications from cell to small animal imaging and even human.16, 17, 18 Dual-dye-labeled Affibody or even tri-labeled Affibody have also been reported.19 Overall, an Affibody protein scaffold has been proven to be an excellent platform for molecular probe development. An anti-EGFR Affibody protein, , has shown strong binding to EGFR with no cross-binding to other growth factor receptors.20 It has been found that - and -labeled display fast and high tumor uptake ( inject dose (ID)/g and , respectively, at p.i.), as well as quick clearance from blood pool ( and , respectively, p.i.) and EGFR nonexpression tissues20, 21, 22, 23, 24 (except kidney). We thus hypothesized that this Affibody could be a good protein for developing optical imaging probes for near-infrared (NIR) fluorescent imaging of EGFR positive tumors. In this research, a cysteine residue was first added to the N-terminus of . The resulting chemically synthesized protein, Ac-Cys- (Ac-CVDNKFNKEMWAAWEEIRNLPNLNGWQMTAFIASLVDDPSQSANLLAEAKKLNDAQAPK- ), was site specifically conjugated with a NIR dye, Cy5.5-mono-maleimide. The optical probe Cy5.5- was further evaluated in the EGFR positive human epithelial carcinoma A431 cell culture and tumor-bearing mice. 2.Materials and Methods2.1.General Material and ReagentCy5.5-mono-maleimide was purchased from GE Healthcare (Piscataway, New Jersey). Dichloromethane, triethylamine, N-hydroxybenzotriazole hydrate (HOBT), diisopropylcarbodiimide (DIC), ethyl acetate, and dithiothriotol (DTT) were purchased from Sigma-Aldrich Chemical Co. (St. Louis, Missouri). Dimethylsulfoxide (DMSO) and ethyl acetate were purchased from Fisher Scientific (Pittsburgh, Pennsylvania). All the other standard reagents were purchased from Sigma-Aldrich Chemical Co. All chemicals were used without further purification. The Affibody analog, Ac-Cys- , was prepared by a solid phase peptide synthesis method as previously described.21 Reverse-phase high-performance liquid chromatography (RP-HPLC) was performed on a Dionex Ultimate 3000 HPLC system (Dionex Corporation, Sunnyvale, California, for analysis and preparation of peptides and bioconjugates) equipped with a photodiode array detector. Five UV wavelengths (218, 254, 280, 500, and ) were monitored for all the experiments. Analytical RP-HPLC column from Sorbent Tech (Atlanta, Georgia) (Grace Vydac C4, ) was used for analysis of the labeled small protein. The mobile phase was solvent A, 0.1% trifluoroacetic acid , and solvent B, 0.1%TFA/acetonitrile. The flow rate was , with the mobile phase starting from 80% solvent A and 20% solvent B to 50% solvent A and 50% solvent B at . Matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS, model: Perseptive Voyager-DE RP Biospectrometer) (Framingham, Massachusetts) or an electrospray ionization quadrupole mass spectrometer (ESI-MS, model: Micromass ZQ single quadrupole LC-MS) (Milford, Massachusetts) was performed by the Stanford Protein and Nucleic Acid Biotechnology Facility and Stanford Chemistry Department Mass Spectrometry Facility, respectively. The human epithelial carcinoma cancer cell line A431 was obtained from the American Type Tissue Culture Collection (Manassas, Virginia). Female athymic nude mice (nu/nu) were purchased from Charles River Laboratories (Boston, Massachusetts). 2.2.Synthesis of Cy5.5-The general procedure for preparation of Cy5.5- is as follows. The Affibody molecule Ac-Cys- was dissolved in freshly degassed phosphate buffer ( , pH 7.4) at a concentration of approximately . The NIR dye Cy5.5-mono-maleimide in DMSO was then added (1.5 equivalents per equivalent of the Affibody). After vortexing for , the reaction mixture was purified by the HPLC equipped with a Grace Vydac 214TP54-C4 column. Characterization of Cy5.5- was confirmed using MALDI-TOF-MS. The purity of the bioconjugate was confirmed by the analytical HPLC. Fluorescence emission of Cy5.5- was measured on the Fluomax-3 fluorophotometer (HORIBA Jobin Yvon, Edison, New Jersey), and the spectrum was scanned from with an increment of . The wavelength of excitation light is . The absorption spectrum of Cy5.5- was recorded on an Agilent 8453 UV-visible ChemStation (Agilent Technologies, Wilmington, Delaware). The spectrum was scanned from with an increment of . 2.3.Fluorescence Microscopy StudyThe A431 and MCF7 cells were cultured in high-glucose Dulbecco modified eagle medium (DMEM) and modified eagle medium (MEM) respectively supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin (Invitrogen Life Technologies, Carlsbad, California). The cell line was maintained in a humidified atmosphere of 5% at , with the medium changed every other day. A confluent monolayer was detached with trypsin and dissociated into a single cell suspension for further cell culture. For fluorescence microscopy study, A431 or MCF7 cells were cultured on the MatTek glass-bottom culture dishes (Ashland, Massachusetts). After , the cells were washed with phosphate-buffered saline (PBS) and then incubated at with Cy5.5- for in dark. The EGFR-binding specificity of Cy5.5- in cell culture was verified by incubating the A431 and MCF7 cells with or without large amounts of (blocking dose) nonfluorescent Ac-Cys- peptide . After the incubation period, cells were washed three times with ice-cold PBS. The fluorescence signal of the cells was recorded using an Axiovert 200M fluorescence microscope (Carl Zeiss MicroImaging, Inc., Thornwood, New Jersey) equipped with a Cy5.5 filter set (Exciter, HQ ; Emitter, HQ ). An AttoArc HBO microscopic illuminator was used as a light source for fluorescence excitation. Images were taken using a thermoelectrically cooled charge-coupled device (CCD) (Micromax, model RTE/CCD-576, Princeton Instruments Inc., Trenton, New Jersey) and analyzed using MetaMorph Software version 6.2r4 (Molecular Devices Corporation, Downingtown, Pennsylvania). 2.4.Binding Affinity Measurement with Flow CytometryThe flow cytometric analysis was performed as described in the literature with minor modification.23 Samples were illuminated with a sapphire laser at and a red laser at on a BD FACSCalibur. The fluorescence and the forward-scattered and side-scattered light from 10,000 cells was detected at a rate of approximately . Flow cytometric data were analyzed with FACSDiva Software (BD Biosciences, San Jose, California). Prior to the flow cytometric analysis, A431 cells seeded in flasks approximately before the experiment were trypsinized [0.05% trypsin to 0.02% ethylenediaminetetraacetic acid (EDTA), , , ; Lonza Verviers S.P.R.L.] followed by a centrifugation ( , ). The pellet was subsequently resuspended in the binding buffer [PBS containing 1% bovine serum albumin (BSA) and 0.1% sodium azide (NaAz)]. A431 cells ( each, expressing receptors/cell) were then distributed in Falcon tubes . After centrifugation, the cells were incubated with of Cy5.5- at different concentrations (ranging from ) for at room temperature in the dark, with continuous rocking. The cells were then centrifuged ( , , ), washed with the binding buffer twice, resuspended in of binding buffer, and subjected to flow cytometric analysis. All procedures after the incubation with the probe were performed on ice with ice-cold buffers. A triplicate of the flow cytometric data was analyzed with GraphPad Prism 5 (GraphPad Software, San Diego, California) and a nonlinear regression one-site-specific model was used to calculate the of the probe. 2.5.Optical Imaging of A431 Tumors in MiceAll animal studies were carried out according to a protocol approved by the Stanford University Administrative Panels on Laboratory Animal Care (APLAC). Female athymic nude mice (nu/nu) at of age were injected subcutaneously in the shoulder with A431 or MCF7 cells suspended in of PBS. For the MCF7 mice model, of estrogen was planted in the back before inoculation. The tumor-bearing mice were subjected to in vivo optical imaging studies after implantation. In vivo fluorescence imaging was performed with an IVIS 200 small animal imaging system (Xenogen, Alameda, California). A Cy5.5 filter set (excitation ; emission ) was used for acquiring Cy5.5- fluorescence in vivo. Identical illumination settings (lamp voltage, filters, /stop, fields of view, binning) were used to acquire all images, and fluorescence emission was normalized to photons per second per centimeter squared per steradian . Images were acquired and analyzed using Living Image 3.0 software (Xenogen, Alameda, California). For the positive control experiment, mice were injected via tail vein with of Cy5.5- and subjected to optical imaging at various time points p.i. For the blocking experiment, mice ( for each probe also) were injected with the mixture of of Ac-Cys- and of Cy5.5- . IVIS-200 NIR fluorescent images were acquired using a exposure time ( , ). To subtract the autofluorescence, optical imaging using an IVIS spectrum small animal imaging system was also performed on mice for ex vivo imaging. The mice injected with Cy5.5- were euthanized at p.i. and used for ex vivo fluorescence imaging. The tumor and other major tissues were dissected and placed on black papers. A slightly blue shifted excitation filters at was used to measure the tissue autofluorescence. The background filter image was then subtracted from the primary filter image (excitation filter ) of Cy5.5- fluorescence by image-math. IVIS-spectrum NIR fluorescent images were acquired using a exposure time ( , ) because of the much narrower emission filter range . The fluorescence images were acquired, and mean fluorescence flux for each sample was obtained. 2.6.Statistical AnalysisAll the data are given as (standard deviation) of independent measurements. Statistical analysis was performed using a Student’s test. Statistical significance was assigned for values . To determine tumor contrast, mean fluorescence intensities of the tumor area at the shoulder of the animal and of the area at the right flank (normal tissue) were calculated by the region-of-interest (ROI) function of Living Image software integrated with Igor (Wavemetrics, Lake Oswego, Oregon). Dividing by yielded the contrast between tumor tissue and normal tissue. 3.Results3.1.Synthesis of Cy5.5-The Affibody small protein Ac-Cys- with a cysteine at the terminal was successfully synthesized using a peptide synthesizer and purified by semipreparative HPLC. The peptide was generally obtained in 10% yield with over 95% purity [Fig. 1 ]. The purified Ac-Cys- was characterized by MALDI-TOF-MS. The measured molecular weight (MW) was consistent with the expected MW: for (Calculated ). Ac-Cys- was then conjugated with Cy5.5-mono-maleimide in the PBS buffer and purified by HPLC. MS analysis verified the success preparation of the final product Cy5.5- (the observed MW: for , calculated ). The purity for the final product was over 95% (retention time: ) [Fig. 1], and the recovery yield of the probe was 47%. Spectrum study showed that Cy5.5- had similar absorption and fluorescence emission characteristics of free Cy5.5 dye, with and [Fig. 1]. 3.2.Binding Specificity and Affinity of Cy5.5-To demonstrate the EGFR specificity and subcellular localization of Cy5.5- , the probe was incubated with A431 tumor cells and fluorescent microscopic imaging was performed. With an incubation at , intensive fluorescent signal was observed from the A431 cells membrane, and some of probe was also found to be inside of the cells (Fig. 2 ). Furthermore, the fluorescent signal from the cells could be significantly reduced by incubation of the cells with large excess of the Ac-Cys- , demonstrating the binding specificity of Cy5.5- (Fig. 2). The staining of low EGFR expression MCF7 is barely visible. The flow cytometry study was performed for measurement of the binding affinity of Cy5.5- . A representative flow cytometric analysis was shown in Fig. 3 . The equilibrium dissociation constant was determined to be using a nonlinear regression one-site-specific model (Fig. 3). 3.3.In Vivo and Ex Vivo Optical ImagingFigure 4 shows typical NIR fluorescent images of nude mice bearing subcutaneous A431 and MCF7 tumor after intravenous injection of of Cy5.5- . The A431 tumor could be clearly visualized from the surrounding background tissue from p.i.. Quantification analysis of ROIs was performed and the tumor to normal tissue ratio as a function of time is depicted in Fig. 4. Cy5.5- exhibited a fast tumor targeting property in vivo. The tumor-to-normal tissue ratio reached a peak at p.i. The receptor specificity of the probe was verified by the blocking experiment. Unlabeled Ac-Cys- significantly reduced tumor uptake and tumor contrast at all the time points [Figure 4]. Tumor contrast as quantified by ROI analysis of images indicated that the value at p.i. was reduced from to . At p.i., MCF7 tumor showed much lower value . Fig. 4(a) In vivo fluorescence IVIS-200 imaging of subcutaneous A431 and MCF7 tumor-bearing nude mice at , , , , and . Cy5.5- with (bottom) or without (top) coinjection of unlabeled Affibody were injected. Arrows indicate the location of tumors. (b) ROI analysis of tumor-to-normal tissue ratios of Cy5.5- in mice bearing A431 tumor at p.i. . 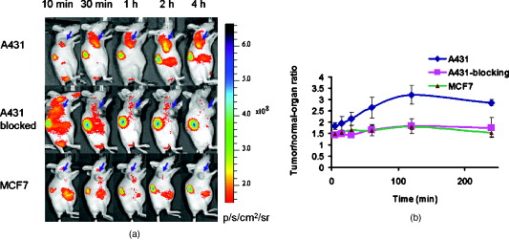 The result for ex vivo tumor mice imaging at p.i., as shown in Fig. 5 . Quantitative analysis showed that there is also large amount of probe trapped inside the kidney without clearance [Fig. 5]. The tumor-to-normal organ (except kidney) ratios are also high [Fig. 5]. For example, the ratios for tumor-to-lung and tumor-to-muscle are and , respectively. Fig. 5(a) Ex vivo imaging of tumor and normal tissues of Cy5.5- after sacrificed the mice at p.i.: Tm, tumor; H, heart; L, lung; Lv, liver; Kn, kidney; Sp, spleen; St, stomach; B, blood; Sk, skin; Ms, Muscle. (B) ROI analysis of fluorescent signal from tumor and normal tissues. (b) Fluorescence intensity ratios of tumor-to-normal tissues based on the ROI analysis. Error bar was calculated as the standard deviation . 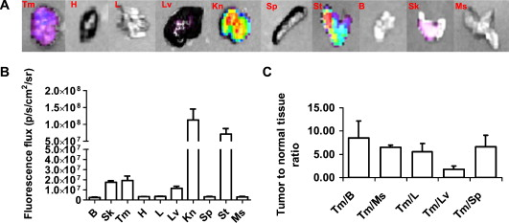 4.DiscussionNIR fluorescent imaging, which uses neither ionizing radiation nor radioactive materials, is emerging as a promising modality that can complement traditional nuclear imaging methods. Optical molecular probes with high sensitivity, stability, fast targeting, and rapid clearance could be prepared.17, 24, 25 Furthermore, optical imaging has very high resolution and shows great potential in clinical translation because of (1) the easy visualization of targeted tissues and (2) its use as supplementary visual aide to provide guidance in surgery.26 Anti-EGFR Affibody, , was radiolabeled with in our recent study, which demonstrated good and fast tumor uptake and long retention.21 It is also of a great interest to us to apply this Affibody molecule for developing an optical imaging probe, since there is a high demand for an EGFR-targeted optical probe with an ideal in vivo imaging profile. To achieve this goal, Affibody analog Cys- was synthesized to render site-specific labeling using maleimide chemistry. Conjugation of the small protein with Cy5.5-mono-maleimide was successfully achieved, moreover the absorption and emission fluorescence properties of the probe remain very close to the unconjugated dye (Fig. 1). The probe Cy5.5- also showed good binding affinity to the A431 cell surface EGFR with a value of , which is considered sufficient for further in vivo evaluation, although target avidity was lower than that of the unmodified Affibody 20 [Fig. 3]. This result implies that the Affibody protein tolerates some terminal modifications. Fluorescent microscopy of Cy5.5- showed that the majority of the probe bond to cell surface receptors after incubation, indicating the slow internalization of the Affibody molecule, which is also consistent with previous finding of the radioactive counterpart.20, 21 More importantly, the specificity of the probe was verified with blocking by unlabeled Affibody (Fig. 2). These results warranted the further evaluation of the probe for in vivo EGFR-targeted tumor imaging. Considering the high sensitivity of in vivo NIR imaging, of Cy5.5- was injected into A431 mice, and optical imaging studies were performed using IVIS-200 systems. Consistent with our expectation, good tumor uptakes and imaging qualities were observed [Fig. 4]. The A431 tumor was clearly delineated from normal tissues at as early as p.i. Quantification analysis of IVIS-200 images illustrated that tumor uptake reach plateau at [Fig. 4]. These results were attributable to the fast tumor targeting and rapid blood clearance abilities of Affibody molecules. Kidney uptake was also very high even at p.i., which is consisted with the radiometal labeled Affibody probes.20, 21, 27 Further ex vivo imaging study exhibited that tumor-to-blood ratio reached at p.i. Autofluorescence was very obvious for the skin, which increases the background. Ex vivo imaging also revealed that tumor-to-muscle ratio was as high as . These properties are favorable and demonstrate the use of the optical probe for intraoperative optical imaging. In vivo optical imaging of EGFR has extensively been investigated using monoclonal antibody such as cetuximab (Erbitux) labeled with NIR dyes.8, 9, 28 Cy5.5-labeled cetuximab (abbreviated as Cy5.5-Erbitux) was reported to target EGFR-high-expression MDA-MB-231 tumors and reached a maximum tumor to normal tissue ratio of ca 3 at p.i.. However, because of the slow localization and clearance of the antibody, Cy5.5-Erbitux showed almost no difference between MDA-MB-231 tumor with high EGFR expression and MCF-7 with low expression9 before p.i. In contrast, Cy5.5- demonstrated fast tumor accumulation ( at p.i.) in the A431 model, while accumulation was very low in MCF7 tumor ( at p.i., ), which is advantageous for both preclinical and clinical applications. NIR-dye-labeled EGF protein has also been reported for in vivo imaging.29, 30 However, EGF as a natural ligand has some side effects such as stimulation of EGFR phosphorylation, causing vomiting and diarrhea.30, 31 Finally, quantum dot conjugated with reduced EGF (r-Egf) was also reported, but the imaging result was poor and toxicity of quantum dot was a concern even at very low amount.32 AcknowledgmentsThis work was supported, in part, by the California Breast Cancer Research Program 14IB-0091 (Z.C.) and an SNM Pilot Research Grant (to Z.C.). We also would like to thank Steven Jing for the help with Affibody synthesis and purification and Edwin Chang for helpful discussions. ReferencesF. X. Real, W. J. Rettig, P. G. Chesa, M. R. Melamed, L. J. Old, and J. Mendelsohn,
“Expression of epidermal growth factor receptor in human cultured cells and tissues,”
Cancer Res., 46
(9), 4726
–4731
(1986). 0008-5472 Google Scholar
E. Van Cutsem, C. H. Köhne, E. Hitre, J. Zaluski, C. R. Chang Chien, A. Makhson, G. D’Haens, T. Pintér, R. Lim, G. Bodoky, J. K. Roh, G. Folprecht, P. Ruff, C. Stroh, S. Tejpar, M. Schlichting, J. Nippgen, and P. Rougier,
“Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer,”
N. Engl. J. Med., 360
(14), 1408
–1417
(2009). https://doi.org/10.1056/NEJMoa0805019 0028-4793 Google Scholar
J. G. Paez, P. A. Jänne, J. C. Lee, S. Tracy, H. Greulich, S. Gabriel, P. Herman, F. J. Kaye, N. Lindeman, T. J. Boggon, K. Naoki, H. Sasaki, Y. Fujii, M. J. Eck, W. R. Sellers, B. E. Johnson, and M. Meyerson,
“EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy,”
Science, 304
(5676), 1497
–1500
(2004). https://doi.org/10.1126/science.1099314 0036-8075 Google Scholar
M. H. Cohen, J. R. Johnson, Y. F. Chen, R. Sridhara, and R. Pazdur,
“FDA drug approval summary: erlotinib (Tarceva) tablets,”
Oncologist, 10 461
–466
(2005). https://doi.org/10.1634/theoncologist.10-7-461 1083-7159 Google Scholar
H. Su, Y. Seimbille, G. Z. Ferl, C. Bodenstein, B. Fueger, K. J. Kim, Y. T. Hsu, S. M. Dubinett, M. E. Phelps, J. Czernin, and W. A. Weber,
“Evaluation of as a molecular imaging probe for the assessment of the epidermal growth factor receptor status in malignant tumors,”
Eur. J. Nucl. Med. Mol. Imaging, 35
(6), 1089
–1099
(2008). https://doi.org/10.1007/s00259-007-0636-6 1619-7070 Google Scholar
A. Pal, A. Glekas, M. Doubrovin, J. Balatoni, T. Beresten, D. Maxwell, S. Soghomonyan, A. Shavrin, L. Ageyeva, R. Finn, S. M. Larson, W. Bornmann, and J. G. Gelovani,
“Molecular imaging of EGFR kinase activity in tumors with -labeled small molecular tracer and positron emission tomography,”
Mol. Imaging Biol., 8
(5), 262
–277
(2006). https://doi.org/10.1007/s11307-006-0049-0 1536-1632 Google Scholar
H. Wang, J. Yu, G. Yang, X. Song, X. Sun, S. Zhao, and D. Mu,
“Assessment of -labeled-4-N-(3-bromoanilino)-6,7-dimethoxyquinazoline as a positron emission tomography agent to monitor epidermal growth factor receptor expression,”
Cancer Sci., 98
(9), 1413
–1416
(2007). https://doi.org/10.1111/j.1349-7006.2007.00562.x Google Scholar
J. P. Gleysteen, J. R. Newman, D. Chhieng, A. Frost, K. R. Zinn, and E. L. Rosenthal,
“Fluorescent labeled anti-EGFR antibody for identification of regional and distant metastasis in a preclinical xenograft model,”
Head Neck, 30
(6), 782
–789
(2008). https://doi.org/10.1002/hed.20782 1043-3074 Google Scholar
K. Wang, K. Wang, W. Li, T. Huang, R. Li, D. Wang, B. Shen, and X. Chen,
“Characterizing breast cancer xenograft epidermal growth factor receptor expression by using near-infrared optical imaging,”
Acta Radiol., 11 1
–9
(2009). 0284-1851 Google Scholar
C. R. Divgi, S. Welt, M. Kris, F. X. Real, S. D. J. Yeh, R. Gralla, B. Merchant, S. Schweighart, M. Unger, S. M. Larson, and J. Mendelsohn,
“Phase I and imaging trial of indium-111-labeled anti-epidermal growth factor receptor monoclonal antibody 225 in patients with squamous cell lung carcinoma,”
J. Natl. Cancer. Inst. (Bethesda), 83
(2), 97
–104
(1991). https://doi.org/10.1093/jnci/83.2.97 Google Scholar
R. J. Hosse, A. Rothe, and B. E. Power,
“A new generation of protein display scaffolds for molecular recognition,”
Protein Sci., 15 14
–27
(2006). https://doi.org/10.1110/ps.051817606 0961-8368 Google Scholar
H. K. Binz, P. Amstutz, and A. Pluckthun,
“Engineering novel binding proteins from nonimmunoglobulin domains,”
Nat. Biotechnol., 23
(10), 1257
–1268
(2005). https://doi.org/10.1038/nbt1127 1087-0156 Google Scholar
K. Nord, E. Gunneriusson, J. Ringdahl, S. Ståhl, M. Uhlén, and P. Å. Nygren,
“Binding proteins selected from combinatorial libraries of an alpha-helical bacterial receptor domain,”
Nat. Biotechnol., 15
(8), 772
–777
(1997). https://doi.org/10.1038/nbt0897-772 1087-0156 Google Scholar
A. Orlova, V. Tolmachev, R. Pehrson, M. Lindborg, T. Tran, M. Sandström, F. Y. Nilsson, A. Wennborg, L. Abrahmsén, and J. Feldwisch,
“Synthetic affibody molecules: a novel class of affinity ligands for molecular imaging of HER2-expressing malignant tumors,”
Cancer Res., 67
(5), 2178
–2186
(2007). https://doi.org/10.1158/0008-5472.CAN-06-2887 0008-5472 Google Scholar
V. Tolmachev, A. Orlova, R. Pehrson, J. Galli, B. Baastrup, K. Andersson, M. Sandström, D. Rosik, J. Carlsson, H. Lundqvist, A. Wennborg, and F. Y. Nilsson,
“Radionuclide therapy of HER2-positive microxenografts using a 177Lu-labeled HER2-specific Affibody molecule,”
Cancer Res., 67
(6), 2773
–2782
(2007). https://doi.org/10.1158/0008-5472.CAN-06-1630 0008-5472 Google Scholar
Z. Cheng, O. P. De Jesus, M. Namavari, A. De, J. Levi, J. M. Webster, R. Zhang, B. Lee, F. A. Syud, and S. S. Gambhir,
“Small-animal PET imaging of human epidermal growth factor receptor type 2 expression with site-specific 18F-labeled protein scaffold molecules,”
J. Nucl. Med., 49
(5), 804
–813
(2008). https://doi.org/10.2967/jnumed.107.047381 0161-5505 Google Scholar
S. B. Lee, M. Hassan, R. Fisher, O. Chertov, V. Chernomordik, G. Kramer-Marek, A. Gandjbakhche, and J. Capala,
“Affibody molecules for in vivo characterization of HER2-positive tumors by near-infrared imaging,”
Clin. Cancer Res., 14
(12), 3840
–3849
(2008). https://doi.org/10.1158/1078-0432.CCR-07-4076 1078-0432 Google Scholar
R. P. Baum, A. Orlova, V. Tolmachev, and J. Feldwisch,
“Receptor PET/CT and SPECT using an Affibody molecule for targeting and molecular imaging of HER2-positive cancer in animal xenografts and human breast cancer patients [abstract],”
J. Nucl. Med., 47
(Supp. l), 108P
(2006). 0161-5505 Google Scholar
T. Engfeldt, B. Renberg, H. Brumer, P. Å. Nygren, and A. E. Karlström,
“Chemical synthesis of triple-labelled three-helix bundle binding proteins for specific fluorescent detection of unlabelled protein,”
ChemBioChem, 6
(6), 1043
–1050
(2005). https://doi.org/10.1002/cbic.200400388 1439-4227 Google Scholar
M. Friedman, A. Orlova, E. Johansson, T. L. Eriksson, I. Höidén-Guthenberg, V. Tolmachev, F. Y. Nilsson, and S. Ståhl,
“Directed evolution to low nanomolar affinity of a tumor-targeting epidermal growth factor receptor-binding Affibody molecule,”
J. Mol. Biol., 376
(5), 1388
–1402
(2008). https://doi.org/10.1016/j.jmb.2007.12.060 0022-2836 Google Scholar
Z. Miao, G. Ren, H. Liu, L. Jiang, and Z. Cheng,
“Small-animal PET imaging of human epidermal growth factor receptor positive tumor with a labeled affibody protein,”
Bioconjugate Chem., 21
(5), 947
–954
(2010). https://doi.org/10.1021/bc900515p Google Scholar
Z. Miao, G. Ren, H. Liu, L. Jiang, S. S. Gambhir, and Z. Cheng,
“A protein scaffold based molecule for EGFR PET imaging,”
J. Nucl. Med., 50
(2), 386
(2009). 0161-5505 Google Scholar
V. Tolmachev, M. Friedman, M. Sandström, T. L. Eriksson, D. Rosik, M. Hodik, S. Ståhl, F. Y. Frejd, and A. Orlova,
“Affibody molecules for epidermal growth factor receptor targeting in vivo: aspects of dimerization and labeling chemistry,”
J. Nucl. Med., 50
(2), 274
–283
(2009). https://doi.org/10.2967/jnumed.108.055525 0161-5505 Google Scholar
T. Troy, D. Jekic-McMullen, L. Sambucetti, and B. Rice,
“Quantitative comparison of the sensitivity of detection of fluorescent and bioluminescent reporters in animal models,”
Mol. Imaging, 3
(1), 9
–23
(2004). https://doi.org/10.1162/153535004773861688 1535-3508 Google Scholar
Z. Cheng, Y. Wu, Z. Xiong, S. S. Gambhir, and X. Chen,
“Near-infrared fluorescent RGD peptides for optical imaging of integrin expression in living mice,”
Bioconjugate Chem., 16
(6), 1433
–1441
(2005). https://doi.org/10.1021/bc0501698 1043-1802 Google Scholar
A. Nakayama, F. del Monte, R. J. Hajjar, and J. V. Frangioni,
“Functional near-infrared fluorescence imaging for cardiac surgery and targeted gene therapy,”
Mol. Imaging, 1
(4), 365
–77
(2002). https://doi.org/10.1162/153535002321093972 1535-3508 Google Scholar
Z. Cheng, O. P. De Jesus, D. J. Kramer, A. De, J. M. Webster, O. Gheysens, J. Levi, M. Namavari, S. Wang, J. M. Park, R. Zhang, H. Liu, B. Lee, F. A. Syud, and S. S. Gambhir,
“-labeled Affibody molecules for imaging of HER2 expressing tumors,”
Mol. Imaging Biol., 12 316
–324
(2009). https://doi.org/10.1007/s11307-009-0256-6 1536-1632 Google Scholar
K. P. Withrow, J. R. Newman, J. B. Skipper, J. P. Gleysteen, J. S. Magnuson, K. Zinn, and E. L. Rosenthal,
“Assessment of bevacizumab conjugated to Cy5.5 for detection of head and neck cancer xenografts,”
Technol. Cancer Res. Treat., 7
(1), 61
–66
(2008). 1533-0346 Google Scholar
S. Ke, X. Wen, M. Gurfinkel, C. Charnsangavej, S. Wallace, E. M. Sevick-Muraca, and C. Li,
“Near-infrared optical imaging of epidermal growth factor receptor in breast cancer xenografts,”
Cancer Res., 63
(22), 7870
–7875
(2003). 0008-5472 Google Scholar
K. E. Adams, S. Ke, S. Kwon, F. Liang, Z. Fan, Y. Lu, K. Hirschi, M. E. Mawad, M. A. Barry, and E. M. Sevick-Muraca,
“Comparison of visible and near-infrared wavelength-excitable fluorescent dyes for molecular imaging of cancer,”
J. Biomed. Opt., 12
(2), 024017
(2007). https://doi.org/10.1117/1.2717137 1083-3668 Google Scholar
A. Cuartero-Plaza, E. Martínez-Miralles, R. Rosell, C. Vadell-Nadal, M. Farré, and F. X. Real,
“Radiolocalization of squamous lung carcinoma with -labeled epidermal growth factor,”
Clin. Cancer Res., 2
(1), 13
–20
(1996). 1078-0432 Google Scholar
P. Diagaradjane, J. M. Orenstein-Cardona, N. E. Colón-Casasnovas, A. Deorukhkar, S. Shentu, N. Kuno, D. L. Schwartz, J. G. Gelovani, and S. Krishnan,
“Imaging epidermal growth factor receptor expression in vivo: pharmacokinetic and biodistribution characterization of a bioconjugated quantum dot nanoprobe,”
Clin. Cancer Res., 14
(3), 731
–741
(2008). https://doi.org/10.1158/1078-0432.CCR-07-1958 1078-0432 Google Scholar
|