|
|
1.IntroductionOuabain is a member of one of the oldest class of pharmaceuticals used for treatment of heart failure,1 and its actions have been revisited since ouabain was identified in 1999 as an endogenous hormone.2 Ouabain is a well-known inhibitor of the -ATPase (sodium/potassium-adenosine triphosphatase; i.e., sodium pump) pathway, an energy-transducing ion pump which catalyzes active transport of sodium and potassium across the plasma membranes,3 and is highly conserved in all eukaryotic cells, including heart cells. Extensive clinical observations demonstrated that in addition to its effects on ionic homeostasis, ouabain also stimulates myocardial metabolism. More specifically, ouabain increases lactic acid utilization by the myocardium, reduces lactic acid concentration in the blood of patients with heart disease, stimulates fatty acid utilization in the myocardium, and inhibits increased oxygen consumption induced by adrenaline.1 Studies over the past few years have clearly demonstrated that ouabain acts by binding on -ATPase, which is an important signal transducer.4 Binding of ouabain to -ATPase changes the interaction of the enzyme with neighboring membrane proteins, and induces the formation of multiple signaling modules, including increased production of reactive oxygen species (ROS). In cardiac myocytes, sodium pump inhibition is known to regulate cardiomyocyte metabolism, including the Ras-Rac-NAD(P)H oxidase cascade5 and redox potential.6 Regulation by ouabain of the sodium pump as an energy transducer thus became an important new line of investigation. Despite the importance of the effects of ouabain on myocardial metabolism, the precise mechanism of its action, particularly its action on the metabolic state of cardiac cells, is still poorly understood. To evaluate the role of ouabain on the mitochondrial metabolic state, we have chosen a noninvasive spectroscopy method based on measuring the time- and spectrally resolved NAD(P)H endogenous fluorescence.7 This technique allows not only to determine changes in the fluorescence intensity, but also to separate individual NAD(P)H fluorescence components accordingly to their spectral characteristics and fluorescence lifetimes.8 Consequently, the applied approach allows us to evaluate changes in the cell’s metabolic oxidative mitochondrial redox state which correlate with redox potential.9,10 This procedure is tested as a perspective pharmaceutical procedure for monitoring the effect of drugs on the mitochondrial metabolism where ouabain is chosen as a model drug. 2.Methods2.1.ChemicalsPyruvate () and 3-β-hydroxybutyrate (BHB, ) were freshly prepared, while lactate () and acetoacetate (AcAc, or 1. ) were added to basic external solution from stock solution (, pH 7.2 for lactate, and 250 mM stock solution for AcAc). Rotenone () and 9,10-dinitrophenol (DNP, ) were added to cells in basic external solution for 5 to 25 min prior to recording. All chemicals were purchased from Sigma (Canada). 2.2.Animals and Cell IsolationFemale Sprague-Dawley rats (13 to 14 weeks old, Charles River, Canada) were studied. Left ventricular myocytes were isolated after retrograde perfusion of the heart with proteolytic enzymes.11 All procedures were performed in accordance with the National Institutes of Health (NIH) and Canadian Council on Animal Care (CCAC) guidelines, and evaluated by the local Comité Institutionnel des Bonnes Pratiques sur les Animaux en Recherche (CIBPAR), accredited by the CCAC. Myocytes were maintained in a storage solution at 4°C until used. Only cardiac myocytes showing clearly defined striations and edges were studied. 2.3.Time-Resolved Spectroscopy Measurements of NAD(P)H FluorescenceNAD(P)H fluorescence was measured in isolated cardiac cells using a spectrally resolved time-correlated single photon counting (TCSPC) setup on an Axiovert 200 inverted microscope, as previously described.7,12 Picosecond laser diode BDL-375 (Becker&Hickl, Boston Electronics, USA) with emission at 375 nm was deployed at the 20 MHz repetition rate as an excitation source. All experiments were performed at room temperature. 2.4.SolutionsThe storage solution contained (in ): NaCl, 130.0; KCl, 5.4; , 1.4; , 0.4; creatine, 10.0; taurine, 20.0; glucose, 10.0; and HEPES, 10.0; titrated to pH 7.30 with NaOH. The basic external solution contained (in ): NaCl, 140.0; KCl, 5.4; , 2.0; , 1.0; glucose, 10.0; HEPES, 10.0; adjusted to pH 7.35 with NaOH. Ouabain was prepared freshly from powder and applied 10 min before use at concentrations from 10 to . 2.5.Data AnalysisThe data are reported as means standard error of the means (SEM). Data were collected from at least three different animals per experimental group, and compared by one-way analysis of variance (ANOVA); was considered statistically significant. Individual components of NAD(P)H fluorescence were estimated using principal component analysis (PCA),7 while the linear unmixing approach was applied to separate individual components in the recorded data, as previously reported13,14 Component amplitude for each resolved fluorescence component was calculated from the fluorescence decay kinetics using a monoexponential fitting procedure at the spectral channel with maximum intensity (450 nm). Fluorescence lifetime for each component was estimated in ns together with the component amplitude. Intensity for each component was calculated as []. 3.Results3.1.Effect of Ouabain on the NAD(P)H Fluorescence LevelsIn cardiac cells, principal endogenous indicators of the cellular oxidative metabolism are localized in mitochondria. The endogenous fluorescence generated after excitation with blue/ultraviolet light is mainly resulting from reduced mitochondrial NAD(P)H.15,16 This principal electron donor for electrochemical gradient necessary for ATP generation is used for noninvasive fluorescent probing of the metabolic state. These molecules exist in their free forms, or as cofactors in enzymes of inner mitochondrial membrane, and are involved in the mitochondrial respiratory chain and fatty acid oxidation. The time-resolved spectroscopy is an effective means to establish individual contribution of the two NAD(P)H forms, based on a spectral shift in the NADH spectrum upon binding to enzymes,17 and their distinct fluorescence lifetime. The short NAD(P)H fluorescence lifetime (0.4 ns), is accepted as an indicator of “free” NAD(P)H, while “protein-bound” NAD(P)H was described to have longer lifetimes (approximately 2 ns).18,19 The effect of oubain on the oxidative metabolic state of freshly isolated living cardiac myocytes was investigated by time-resolved spectroscopy of endogenous NAD(P)H fluorescence [see Fig. 1(a)], as described previously.7 A linear unmixing approach was used to separate individual NAD(P)H components; component 1 with fluorescence lifetime of around 1.8 ns corresponded to NAD(P)H in a more viscous environment, and this value is in agreement with published values of “bound” NAD(P)H in mitochondria20,21 [see resolved fluorescence decays in Fig. 1(b)]. The red-shifted component 2 with the fluorescence lifetime of 0.5 to 0.6 ns corresponded to NAD(P)H in a less viscous environment, and this value correlates with published values for “free” NAD(P)H in mitochondria20,21 [Fig. 1(c)]. The residual component 3 was also resolved [Fig. 1(d)], and with the maximum emission between 504 and 520 nm and fluorescence lifetime around 2 ns, it corresponded to flavin fluorescence.8,12 Ouabain had no significant effect on the fluorescence of component 3 at either concentration. Rising concentrations of ouabain affected mainly the integral intensity of component 2 [Fig. 1(c)] due to a drug-related effect on the amplitude of this component [Fig. 2(a), gray circles], which decreased with the dose of ouabain. This effect, observed in the absence of the ouabain-related action on fluorescence lifetimes of the two components [Fig. 2(b)], also had repercussions on decreasing intensity of the component, estimated as component amplitude fluorescence lifetime [Fig. 2(c)]. We noted no significant effect of ouabain on the resolved NAD(P)H component 1 [Fig. 2(a)–2(c), black squares]. Fig. 1Effect of ouabain on NAD(P)H fluorescence. (a) Original recording of the time-resolved spectroscopy measurements of NAD(P)H fluorescence intensity in living cardiac cells, excitation 375 nm (mean of 10 measurements; in the inset, representative transmission image of the single cardiomyocyte illumination). Effect of ouabain (100 and 1000 nmol/L) on the integral NAD(P)H fluorescence intensity of (b) component 1, (c) component 2, and (d) component 3, resolved by spectral unmixing. Note that the fluorescence intensity at each spectral channel is calculated as the integral of the time-resolved fluorescence intensity at the corresponding spectral channel. T‘he effect of ouabain on the fluorescence decays of the three components is illustrated at spectral maximum of 450 nm. 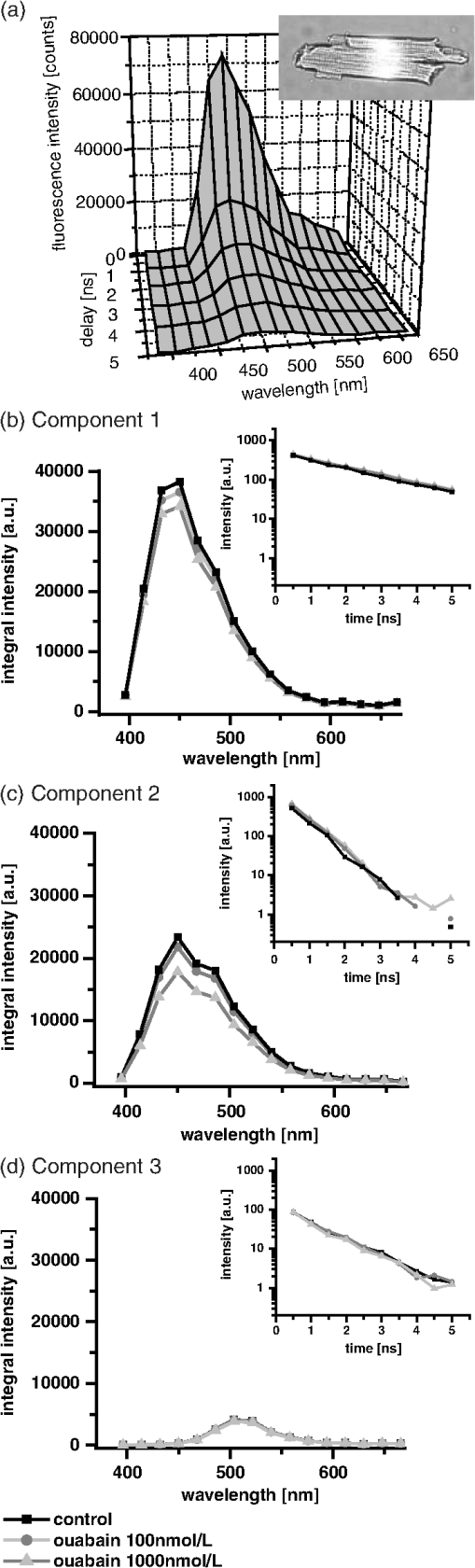 Fig. 2Concentration-dependent effect of ouabain on NAD(P)H fluorescence components. Comparison of the effect of rising concentrations of ouabain on (a) component amplitude and (b) fluorescence lifetime of the NAD(P)H fluorescence components 1 (black squares) and 2 (gray circles). (c) The relationship between the intensity of the component, estimated as: component amplitude fluorescence lifetime. Ouabain concentration is also evaluated (control, ; ouabain 100 nmol/L, ; ouabain 300 nmol/L to 2500 nmol/L, ); versus control. 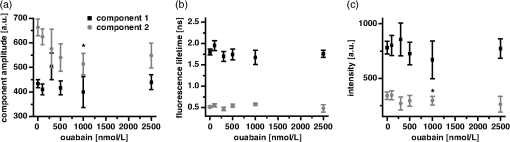 3.2.Effect of Ouabain on Cell Oxidation LevelsPercentage of oxidized nucleotides is an important indicator of the state of the cell oxidation. To understand the implications of the modification of metabolic oxidative state by ouabain, we evaluated the effect of the drug on the percentage of oxidized nucleotides. Rotenone (), the inhibitor of Complex I in the respiratory chain,22,23 was used to put the cell in a fully reduced state at distinct concentrations of the glycoside, while DNP (), an uncoupling agent for ATP synthesis,16 was applied to induce a fully oxidized state [see an example of the actions of rotenone and DNP in the presence of ouabain at the component 1 and the component 2 in Fig. 3(a) and 3(b), respectively]. We calculated the percentage of oxidized nucleotides from the integral fluorescence intensity as [(fully reduced) − (control)]/[(fully reduced) − (fully oxidized)], where corresponds to the sum of integral fluorescence intensity for the two resolved NAD(P)H components. Using this approach, we demonstrated a clear decrease in this parameter from 70% in control conditions to about 35% at high concentrations of ouabain, with a half-response at [Fig. 3(c)]. Altogether, gathered data strongly indicate that ouabain induces a marked decrease in the cell’s oxidation levels. Fig. 3Effect of ouabain on oxidized nucleotides. The effect of rotenone (), the inhibitor of the respiratory chain, and 9,10-dinitrophenol (DNP, ), the uncoupler of the respiratory chain, are illustrated on the fluorescence intensity of the NAD(P)H component 1 (a) and 2 (b) in the presence of ouabain (ouabain , ; ouabain in the presence of rotenone or DNP, ). (c) Concentration-dependent effect of ouabain on percentage of oxidized nucleotides is evaluated as [(fully reduced) − (control)]/[(fully reduced) − (fully oxidized)], where the fully reduced state is induced in the presence of rotenone, and the fully oxidized state is induced in the presence of DNP. 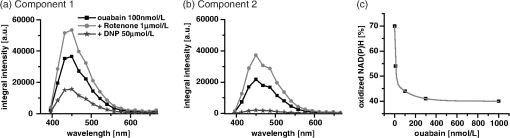 redox potential is the driving force of oxidative phosphorylation, and its increase leads to a linear rise of maximal respiration rate in isolated heart mitochondria.24,25 This potential decreased in cardiac myocytes following ouabain action.6 These findings prompted us to evaluate changes in the cell metabolic oxidative redox state by determination of the mitochondrial ratio in cardiac cells. The ratio of the “free”/“bound” amplitude was proposed to correspond to the reduction/oxidation pair,9,10 and thus is considered an important indicator of modifications in the mitochondrial metabolic status. We evaluated the concentration-dependent effect of ouabain on the component amplitudes ratio of the resolved components, calculated as the component 2 to component 1 amplitude. Increasing concentrations of the glycoside decreased the component amplitudes ratio [Fig. 4(c), black squares]. This result is in accordance with a previously published observation that ouabain decreased the redox potential in cardiac myocytes.6 This result also confirms that ouabain decreases mitochondrial oxidation levels. Fig. 4Effect of lactate and pyruvate on the effect of ouabain on NAD(P)H. The effect of lactate () in the presence of pyruvate () on the component amplitude of the resolved components 1 (a), and 2 (b) at different concentrations of ouabain (lactate and pyruvate alone, ; lactate and pyruvate in the presence of ouabain , ; lactate and pyruvate in the presence of ouabain , ; for numbers in control conditions, see legend of Fig. 2). The effect of ouabain on the integral fluorescence intensity of the two resolved components is shown. (c) Comparison of the effect of ouabain in control conditions (black squares) and in the presence of lactate and pyruvate (gray circles) on the amplitude ratio of component 1. 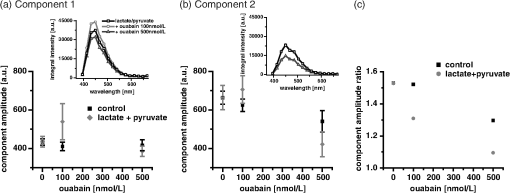 3.3.Effect of Ouabain in the Presence of Lactate and PyruvateOuabain is known to increase lactic acid utilization by the myocardium and reduce lactic acid concentration in the blood of patients with heart disease.1 We have previously demonstrated the importance of the switch in the use of the lactate/pyruvate as an energy substrate as opposed to glucose alone on the cardiomyocyte function in both physiological and pathological situations.11,26 Therefore, we compared the effect of ouabain (100 and ) after application of lactate () in the presence of pyruvate (), both applied in concentrations found in the blood of studied animals,11 to promote the rise in the cytosolic NADH production. Despite little effect on the integral intensity or amplitude of the two resolved NAD(P)H components [Fig. 4(a) and 4(b)], and the observations confirming the primarily mitochondrial origin of the studied fluorescence, lactate and pyruvate stimulated the effect of ouabain on the component amplitudes ratio [Fig. 4(c), gray circles]. This finding points to a possible contribution of cytosolic NADH in the ouabain action. 3.4.Sensitivity of Mitochondrial NADH Production to OuabainTo understand all aspects of the effect of ouabain on mitochondrial metabolic state, we also tested the role that the glycoside plays in the production of NADH by cardiomyocyte mitochondria. In cardiac tissue, NADH is produced by fatty acid oxidation from acetyl-CoA entering the Krebs cycle and this reaction depends on the BHB/AcAc ratio.27 As increasing the BHB/AcAc ratio from to favors the NADH production, we administered BHB () in a basic extracellular solution in the presence of different AcAc concentrations: 1.5 and ( and ratio, respectively) to vary NADH production. In accordance with decreased NADH concentration in cardiomyocyte mitochondria, the lowered ratio translated into a decrease of the integral fluorescence intensity in components 1 and 2 [Fig. 5(a)], due to the smaller amplitude of the two components [Fig. 5(b)], leading to a more important effect of BHB/AcAc on the component amplitudes ratio [Fig. 5(c)]. By inducing a lower increase in the amplitude of the resolved components, ouabain (100 nM) affected the capacity of BHB/AcAc to stimulate the NADH production. Based on the gathered data, maximal production of NADH was calculated from the integral intensity of both components as [control − (BHB:AcAc )]/[(BHB:AcAc ) − (BHB:AcAc )]. In this way, we estimated maximal production of NADH as 55.7% in control cells; in the presence of ouabain (), the NADH production increased to 75.5%, suggesting that ouabain increases the capacity of mitochondria to produce NADH. Fig. 5Effect of ouabain on mitochondrial NADH production. The effect of BHB/AcAc 2:1 (), or BHB/AcAc () in the presence of ouabain (; ouabain alone ) on (a) the fluorescence intensity, and (b) the component amplitude of the two resolved components, as well as (c) the amplitude ratio of component 1. Control conditions (in the absence of ouabain, ), BHB/AcAc in control conditions (), BHB/AcAc in control conditions (); versus control, or versus ouabain. 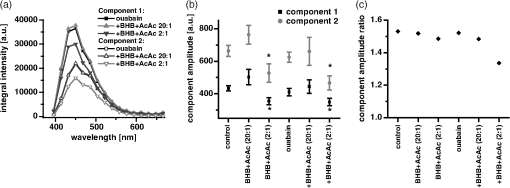 4.DiscussionIn this study, we present a novel approach aimed to evaluate the effect of pharmaceutical drugs on the oxidative metabolic state of living cardiac cells by time-resolved spectroscopy of endogenous NAD(P)H fluorescence, using ouabain as a representative of a widely used class of pharmaceuticals. Our data revealed that the glycoside is capable of affecting cardiomyocyte metabolism. More precisely, 1. ouabain decreased the integral NAD(P)H intensity due to reduced amplitude of the NAD(P)H component 2. leading to a lowered component amplitudes ratio which indicates a change in metabolic oxidative state. 2. Cell oxidation levels, evaluated as percentage of oxidized NAD(P)H, decreased exponentially with rising concentrations of the cardiac glycoside. 3. Lactate/pyruvate, affecting the cytosolic NADH gradient, increased the effect of ouabain on the component amplitudes ratio. 4. Ouabain stimulated the mitochondrial NADH production. Overall, our data point to the fact that ouabain is acting by decreasing cell oxidation in cardiac myocytes. Evaluation of the effects of pharmaceutical drugs on the mitochondrial metabolic state is crucial for understanding their potential role in affecting cellular energetics. Currently there is a lack of noninvasive techniques that would allow monitoring the effect of drugs on the cell metabolism directly in living cells and tissues. In recent years, advanced optical methods were developed for detection of mitochondrial oxidative state in living systems. Latest developments in time-resolved spectroscopy include the multi-photon excited time-resolved fluorescence, as well as spectrally resolved time-correlated single photon counting of various endogenously fluorescing molecules.8,28 Fluorescence lifetime imaging uses tissue autofluorescence (AF) to characterize endogenous fluorophores [NAD(P)H and flavins] which reflect redox processes in the mitochondrial matrix, and thus allow monitoring of their important role in the cell metabolism. Time-resolved spectroscopy measurements29,30 of NAD(P)H and flavins were shown to be suitable for scoring the metabolic state of various cells, and serve as optical biomarkers in cardiomyocytes, but also in other cells including cancer cells and precancerous lesions.8 Additional improvements in the detection by AF can be obtained by separating individual AF components using time-resolved and/or spectral techniques.13,14,31 In this study, we applied time-resolved spectroscopy of endogenous NAD(P)H fluorescence to test a perspective procedure for evaluating the effect of pharmaceutical drugs on the mitochondrial metabolism. Ouabain was chosen as a model drug. Ouabain was shown to affect cardiovascular function,32 as lowering of regional myocardial blood flow altered the drug binding properties and participated in the reduction of cardiac glycoside binding after reperfusion of ischemia myocardium.33 Other researchers demonstrated that ouabain protects rat hearts against ischemia-reperfusion injury via a pathway involving ROS.34 Ouabain was also proposed to regulate mitochondrial metabolism through -ATPase-mediated signal transmission;3 however, there is rather confusing information on the role of cardiac glycosides in the modulation of the mitochondrial metabolism. Our data confirm the capacity of ouabain to affect mitochondrial metabolic state by acting on the amount of NAD(P)H molecules in cardiac cells. Lack of significant effect of the ouabain on the fluorescence lifetimes of NAD(P)H fluorescence indicates that this effect is most likely due to regulation of NAD(P)H production and utilization, without affecting molecular environment. Our data point to the fact that the effect of ouabain is affected by modification in the energy substrates, particularly lactate/pyruvate versus glucose. We have previously demonstrated11 that a switch in the use of these substrates is highly important in certain physiological conditions, such as physiological and pathological pregnancy, which are also characterized by increased levels of endogenous ouabain.35 This result can be of importance also in other pathological conditions as the sodium pump is regulated in conditions such as hyperglycemia associated with diabetes,36 and is linked to mediation of the glucose uptake and/or lactate production. -ATPase was shown to interact with multiple signaling proteins to transmit ouabain signals,3,37 and several intracellular pathways are known to be implicated in this transmission; namely, ouabain was shown to affect mitochondrial NAD(P)H and ROS generation.5 Recently, oxidative signaling based on NAD(P)H oxidase and superoxide was demonstrated to play an important role in the pump inhibition.38 These authors proposed that regulation of the pump is governed by redox signaling via activation of colocalized NAD(P)H oxidase. Oxidative regulation of the pump function is thought to be of pathophysiological importance, particularly in the heart failure. We previously demonstrated that oxidative stress has a repercussion of lowering of the NAD(P)H fluorescence via stimulation of NAD(P)H-dependent pathways (see supplement in Ref. 26). Consequently, we propose the investigation of the ouabain effect on NAD(P)H-dependent pathways should be the target of future studies. At the same time, failure of ouabain to affect the third residual flavin component indicates that its action is not likely to involve modification in the flavoprotein complexes, but rather a drug-related action on the amount of available NAD(P)H molecules in the cell. Overall, our data demonstrate that, in cardiomyocytes, ouabain exercises a balanced effect on decreasing the cell oxidation levels and increasing the NADH production. 5.ConclusionsOur study uncovers the role of ouabain in the regulation of the metabolic oxidative state, and presents a perspective approach for pharmaceutical testing of drug-related effects on the mitochondrial metabolism by means of time-resolved spectroscopy of endogenous NAD(P)H fluorescence. Gathered data point to the fact that ouabain provokes a change of the metabolic oxidative state towards lowering of the cellular oxidation levels. Ouabain decreases the percentage of oxidized nucleotides while stimulating the NADH production in mitochondria, pointing to a carefully balanced effect of the drug on cellular energetics. To our knowledge, this study is the first to examine the effect of ouabain on the oxidative metabolic state in living cardiac myocytes by evaluating the NAD(P)H fluorescence by lifetime spectroscopy. AcknowledgmentsAuthors acknowledge support from the Heart and Stroke Foundation of Quebec, Integrated Initiative of European Laser Infrastructures LaserLab Europe II (EC’s Seventh Framework Programme FP7/2007-2013 under grant agreement No. 228334), and the Research grant agency of the Ministry of Education, Science, Research and Sport of the Slovak Republic VEGA No. 1/0296/11. F. Elzwiei is a recipient of the Libyan Ministry of Superior Studies. The authors would like to thank V. Bassien-Capsa for help with cell preparation, and C. Des Rosiers for the generous gift of AcAc. ReferencesH. Furstenwerth,
“Ouabain—the insulin of the heart,”
Int. J. Clin. Pract., 64
(12), 1591
–1594
(2010). http://dx.doi.org/10.1111/j.1742-1241.2010.02395.x 1368-5031 Google Scholar
A. Y. BagrovJ. I. ShapiroO. V. Fedorova,
“Endogenous cardiotonic steroids: physiology, pharmacology, and novel therapeutic targets,”
Pharmacol. Rev., 61
(1), 9
–38
(2009). http://dx.doi.org/10.1124/pr.108.000711 PAREAQ 0031-6997 Google Scholar
Z. XieT. Cai,
“--ATPase-mediated signal transduction: from protein interaction to cellular function,”
Mol. Interv., 3
(3), 157
–168
(2003). http://dx.doi.org/10.1124/mi.3.3.157 1534-0384 Google Scholar
Z. Xie,
“Molecular mechanisms of -ATPase-mediated signal transduction,”
Ann. New York Acad. Sci., 986 497
–503
(2003). http://dx.doi.org/10.1111/nyas.2003.986.issue-1 ANYAA9 0077-8923 Google Scholar
Z. Xieet al.,
“Intracellular reactive oxygen species mediate the linkage of -ATPase to hypertrophy and its marker genes in cardiac myocytes,”
J. Biol. Chem., 274
(27), 19323
–19328
(1999). http://dx.doi.org/10.1074/jbc.274.27.19323 JBCHA3 0021-9258 Google Scholar
T. LiuD. A. BrownB. O'Rourke,
“Role of mitochondrial dysfunction in cardiac glycoside toxicity,”
J. Mol. Cell. Cardiol., 49
(5), 728
–736
(2010). http://dx.doi.org/10.1016/j.yjmcc.2010.06.012 JMCDAY 0022-2828 Google Scholar
D. Chorvat Jr.et al.,
“Rejection of transplanted hearts in patients evaluated by the component analysis of multi-wavelength NAD(P)H fluorescence lifetime spectroscopy,”
J. Biophotonics, 3
(10–11), 646
–652
(2010). http://dx.doi.org/10.1002/jbio.201000040 JBOIBX 1864-063X Google Scholar
D. Chorvat Jr.A. Chorvatova,
“Multi-wavelength fluorescence lifetime spectroscopy: a new approach to the study of endogenous fluorescence in living cells and tissues,”
Laser Phys. Lett., 6
(3), 175
–193
(2009). http://dx.doi.org/10.1002/lapl.v6:3 1612-2011 Google Scholar
D. K. Birdet al.,
“Metabolic mapping of MCF10A human breast cells via multiphoton fluorescence lifetime imaging of the coenzyme NADH,”
Cancer Res., 65
(19), 8766
–8773
(2005). http://dx.doi.org/10.1158/0008-5472.CAN-04-3922 CNREA8 0008-5472 Google Scholar
R. Niesneret al.,
“Noniterative biexponential fluorescence lifetime imaging in the investigation of cellular metabolism by means of NAD(P)H autofluorescence,”
Chemphyschem, 5
(8), 1141
–1149
(2004). http://dx.doi.org/10.1002/(ISSN)1439-7641 CPCHFT 1439-4235 Google Scholar
V. Bassien-Capsaet al.,
“Structural, functional and metabolic remodeling of rat left ventricular myocytes in normal and in sodium-supplemented pregnancy,”
Cardiovasc. Res., 69
(2), 423
–431
(2006). http://dx.doi.org/10.1016/j.cardiores.2005.10.017 CVREAU 0008-6363 Google Scholar
D. Chorvat Jr.A. Chorvatova,
“Spectrally resolved time-correlated single photon counting: a novel approach for characterization of endogenous fluorescence in isolated cardiac myocytes,”
Eur. Biophys., 36
(1), 73
–83
(2006). http://dx.doi.org/10.1007/s00249-006-0104-4 EBJOE8 0175-7571 Google Scholar
D. Chorvat Jr.et al.,
“Spectral unmixing of flavin autofluorescence components in cardiac myocytes,”
Biophys. J., 89
(6), L55
–L57
(2005). http://dx.doi.org/10.1529/biophysj.105.073866 BIOJAU 0006-3495 Google Scholar
D. Chorvat Jr.et al.,
“Application of spectral unmixing in multi-wavelength time-resolved spectroscopy,”
Proc. SPIE, 6771 677105
(2007). http://dx.doi.org/10.1117/12.732880 PSISDG 0277-786X Google Scholar
B. Chance,
“Pyridine nucleotide as an indicator of the oxygen requirements for energy-linked functions of mitochondria,”
Circ. Res., 38
(5 Suppl. 1), I31
–I38
(1976). CIRUAL 0009-7330 Google Scholar
D. N. RomashkoE. MarbanB. O'Rourke,
“Subcellular metabolic transients and mitochondrial redox waves in heart cells,”
Proc. Natl. Acad. Sci. USA, 95
(4), 1618
–1623
(1998). http://dx.doi.org/10.1073/pnas.95.4.1618 1091-6490 Google Scholar
B. ChanceJ. B. Neilands,
“Studies on lactic dehydrogenase of heart, II. a compound of lactic dehydrogenase and reduced pyridine nucleotide,”
J. Biol. Chem., 199
(1), 383
–387
(1952). JBCHA3 0021-9258 Google Scholar
D. M. JamesonV. ThomasD. M. Zhou,
“Time-resolved fluorescence studies on NADH bound to mitochondrial malate dehydrogenase,”
Biochim. Biophys. Acta, 994
(2), 187
–190
(1989). http://dx.doi.org/10.1016/0167-4838(89)90159-3 BBACAQ 0006-3002 Google Scholar
J. R. Lakowiczet al.,
“Fluorescence lifetime imaging of free and protein-bound NADH,”
Proc. Natl. Acad. Sci. USA, 89
(4), 1271
–1275
(1992). http://dx.doi.org/10.1073/pnas.89.4.1271 1091-6490 Google Scholar
K. Blinovaet al.,
“Distribution of mitochondrial NADH fluorescence lifetimes: steady-state kinetics of matrix NADH interactions,”
Biochemistry, 44
(7), 2585
–2594
(2005). http://dx.doi.org/10.1021/bi0485124 BICHAW 0006-2960 Google Scholar
H. D. Vishwasraoet al.,
“Conformational dependence of intracellular NADH on metabolic state revealed by associated fluorescence anisotropy,”
J. Biol. Chem., 280
(26), 25119
–25126
(2005). http://dx.doi.org/10.1074/jbc.M502475200 JBCHA3 0021-9258 Google Scholar
K. J. DaviesJ. H. Doroshow,
“Redox cycling of anthracyclines by cardiac mitochondria, I. anthracycline radical formation by NADH dehydrogenase,”
J. Biol. Chem., 261
(7), 3060
–3067
(1986). JBCHA3 0021-9258 Google Scholar
R. S. BalabanF. W. Heineman,
“Control of mitochondrial respiration in the heart in vivo,”
Mol. Cell. Biochem., 89
(2), 191
–197
(1989). http://dx.doi.org/10.1007/BF00220775 MCBIB8 0300-8177 Google Scholar
R. Moreno-Sanchez,
“Regulation of oxidative phosphorylation in mitochondria by external free Ca2+ concentrations,”
J. Biol. Chem., 260
(7), 4028
–4034
(1985). JBCHA3 0021-9258 Google Scholar
V. K. MoothaA. E. AraiR. S. Balaban,
“Maximum oxidative phosphorylation capacity of the mammalian heart,”
Am. J. Physiol., 272
(2 Pt. 2), H769
–H775
(1997). AJPHAP 0002-9513 Google Scholar
V. Bassien-Capsaet al.,
“Metabolic remodelling of cardiac myocytes during pregnancy: the role of mineralocorticoids,”
Can. J. Cardiol., 27
(6), 834
–842
(2011). http://dx.doi.org/10.1016/j.cjca.2011.05.009 CJCAEX 0828-282X Google Scholar
L. H. OpieP. Owen,
“Effects of increased mechanical work by isolated perfused rat heart during production or uptake of ketone bodies. Assessment of mitochondrial oxidized to reduced free nicotinamide-adenine dinucleotide ratios and oxaloacetate concentrations,”
Biochem. J., 148
(3), 403
–415
(1975). BIJOAK 0264-6021 Google Scholar
W. Becker, Advanced Time-Correlated Single Photon Counting Techniques, Springer, New York
(2005). Google Scholar
D. V. O'ConnorD. Phillips, Time-Correlated Single Photon Counting, Academic Press, London
(1984). Google Scholar
J. R. Lakowicz, J. R. Principles of Fluorescence Spectroscopy Introducing the Phase-Modulation Methods, Springer, New York
(1999). Google Scholar
T. ZimmermannJ. RietdorfR. Pepperkok,
“Spectral imaging and its applications in live cell microscopy,”
FEBS Lett., 546
(1), 87
–92
(2003). http://dx.doi.org/10.1016/S0014-5793(03)00521-0 FEBLAL 0014-5793 Google Scholar
P. Manuntaet al.,
“Left ventricular mass, stroke volume, and ouabain-like factor in essential hypertension,”
Hypertension, 34
(3), 450
–456
(1999). http://dx.doi.org/10.1161/01.HYP.34.3.450 Google Scholar
G. A. BellerJ. ConroyT. W. Smith,
“Ischemia-induced alterations in myocardial ()-ATPase and cardiac glycoside binding,”
J. Clin. Invest., 57
(2), 341
–350
(1976). http://dx.doi.org/10.1172/JCI108285 JCINAO 0021-9738 Google Scholar
P. Pasdoiset al.,
“Ouabain protects rat hearts against ischemia-reperfusion injury via pathway involving src kinase, mitoKATP, and ROS,”
Am. J. Physiol., 292
(3), H1470
–H1478
(2007). http://dx.doi.org/10.1152/ajpheart.00877.2006 AJPHAP 0002-9513 Google Scholar
D. A. Lopatinet al.,
“Circulating bufodienolide and cardenolide sodium pump inhibitors in preeclampsia,”
J. Hypertens., 17
(8), 1179
–1187
(1999). http://dx.doi.org/10.1097/00004872-199917080-00018 JOHYD3 0263-6352 Google Scholar
P. A. Hansenet al.,
“Alloxan-induced diabetes reduces sarcolemmal pump function in rabbit ventricular myocytes,”
Am. J. Physiol., 292
(3), C1070
–C1077
(2006). http://dx.doi.org/10.1152/ajpcell.00288.2006 AJPHAP 0002-9513 Google Scholar
Z. XieA. Askari,
“-ATPase as a signal transducer,”
Eur. J. Biochem., 269
(10), 2434
–2439
(2002). http://dx.doi.org/10.1046/j.1432-1033.2002.02910.x EJBCAI 0014-2956 Google Scholar
G. A. Figtreeet al.,
“Reversible oxidative modification: a key mechanism of pump regulation,”
Circ. Res., 105
(2), 185
–193
(2009). http://dx.doi.org/10.1161/CIRCRESAHA.109.199547 CIRUAL 0009-7330 Google Scholar
|

