|
|
1.IntroductionUnderstanding the process of tissue aging is the center of a large research interest which has been strongly focused on investigations on collagen, the most abundant long-life structural protein in humans. It has been demonstrated that during normal aging, type I collagen loses flexibility, becomes stiffer, less soluble and more resistant to enzyme digestion.1 These age-related changes have a direct impact on tissue organization and its biomechanical properties, leading to tissue damages.2 They are mainly due to post-translational modifications such as tissue glycation,3 leading to the formation of the so-called advanced glycation end products (AGEs) implicated in numerous physiopathological complications.4 Nonlinear optical microscopy techniques have been recently introduced to study skin aging using two-photon excited autofluorescence (TPEF) (essentially from AGEs and elastin) and second harmonic generation (SHG) (from collagen). These studies focus essentially on the morphological alterations of the collagen network,5,6 or on scoring indicators (such as the TPEF/SHG intensity ratio) as markers of collagen degradation.7,8 While these analyses are performed at a macroscopic scale, the knowledge of microscopic scale molecular processes is still necessary to better understand the impact of aging on the collagen biomechanical properties. In this work, we investigate the microscopic scale structural behavior of different-age type I collagen isolated fibrils using polarization resolved SHG (PSHG). This technique has been used in tissues;9–11 however, recent works have underlined the complexity of its application in such complex samples, where scattering and birefringence can lead to polarization distortions.12,13 To avoid such complication, we focus our study on a two-dimensional (2-D) model system made of coated type I collagen isolated fibrils submicrometric thickness.14 To analyze PSHG data, a symmetry decomposition of the molecular orientational distribution in collagen fibrils is implemented, which permits to access to local molecular order information in a generic approach without invoking an a priori model for the molecular orientation, as usually done.9–11 2.Materials and MethodsAcid-extracted, native type I collagens were isolated from newborn (four to six days), young adult (two months), six months), and old (two years) rat tail tendons. For each age-group, the number of sacrificed animals had to be adjusted in order to reach the same yield of collagen (1 g): 105 (newborn), 8 (young adult), 7 (adult), and 5 (old). Glycated collagen was obtained by incubating at 4°C for five days the lyophilized collagen () with 3 M of ribose in 0.15 M phosphate buffer pH 7.4. Then collagens were extensively dialyzed against distilled water and lyophilized.15 For 2-D coatings, lyophilized collagens solubilized in 18 mM acetic acid were coated on the surface of a 4-well chambered coverglass Lab-TeK system (Nunc, France), at a concentration of . Then coated substrates were dried overnight at room temperature and rinsed in water.14 The PSHG imaging set-up is based on an inverted two-photon excitation microscope in which the incident wavelength 800 nm from a Ti:Sapphire laser is focused by a high numerical aperture objective (NA 1.15) after reflection on a dichroic mirror. Images are performed by scanning the beam in the sample plane using galvanometric mirrors at a rate of about 1 image per second. The detected SHG signal is filtered at 400 nm and directed toward a photomultiplier. We ascertained that the sample does not affect the incident polarization by scattering or birefringence;12 therefore, an unpolarized detection can be used. Polarization distortions from the optical set-up were also characterized beforehand and included in the signal analysis.12 PSHG consists in rotating the incident linear polarization using a half wave plate on a step motor mount, recording one image per incident polarization between (relative to the horizontal direction in the sample plane) and 180-deg in 32 steps. The data analysis uses a Fourier decomposition of the polarization dependence signal ,11 from which molecular order information is deduced following a methodology described previously.16 In this generic approach, nonlinear active molecules of collagen fibrils are considered to lie within a molecular distribution function which can be reduced to a 2-D function:16 , with the angle between the individual molecule’s axis and the horizontal direction in the sample plane, and the global orientation of the distribution with respect to . The so-called order parameters () are deduced from the normalized Fourier coefficients () by minimizing the mean square error between experimental coefficients and theoretical predictions.16 The quantity is used as an indicator of molecular order, while indicates an asymmetry of the distribution.16 This model is general and does not require an a priori known molecular distribution to quantify molecular order. For the comparison of different age conditions, 20 subregions of the SHG images in each age-group ( size), where the fibrils are the most isolated, were considered. The quantities “SHG efficiencies” and “ values” considered for ANOVA statistical analyses () were averaged over each subregion and represented by their mean and standard error of the mean (SEM). 3.Results and DiscussionSHG intensity images of four different-age coated type I collagens (newborn, young adult, adult, and old) and ribose-glycated collagen are presented in Fig. 1. A direct morphological observation of these collagens, coated at the same concentration, show fibril networks and individual fibrils that become thicker and sparser in young adults compared to newborns. In addition, their length markedly increases with the age of the specimens. Two-dimensional fast Fourier transform (FFT) representations of the corresponding images (Fig. 1) show furthermore that a 1D straight structure of fibrils is highly pronounced for young adults (with visible privileged orientations in the FFT image), less pronounced for adults, and absent in the cases of newborn and old rats. As a comparison, the glycated sample is close to the adult case (Fig. 1). The averaged diameter of the fibrils [Fig. 2(a)], which is above the 300 nm diffraction limit size, shows a similar trend with thicker fibrils for the young adults. Although the glycated sample exhibits a similar morphology as the adults, it is constituted of thicker fibrils. The averaged SHG intensity of the images [Fig. 2(c)] seems to be correlated with the fibril diameters, the higher SHG intensity of thicker fibrils being most probably a signature of their higher collagen content. Fig. 1SHG intensity images of coated type I collagen fibrils (and corresponding 2-D FFT maps shown as insets), as a function of age and for ribose-glycation. Vertical scale: total photon counts summed over 32 incident polarization angles. Image size: μm2 (). Averaged incident power: 10 mW at the sample plane.  Fig. 2(a) Fibril diameter at Full Width Half Maximum (FWHM) as a function of age of the collagen fibrils and for the ribose-glycated collagen ( fibrils were measured per condition, mean and SEM are shown). FWHM are obtained from a fit of Gaussian profiles from single fibrils. (b) Typical cross section of a young adult collagen fibril with Gaussian fit (continuous line, deduced FWHM 400 nm) on a fibril shown in the inset. The white bar corresponds to the profile support. (c) Average intensities (mean and SEM) measured over subregions of the SHG images for each age-group (see text). All conditions were significantly different at 1% level (One way ANOVA, Tukey HSD test). 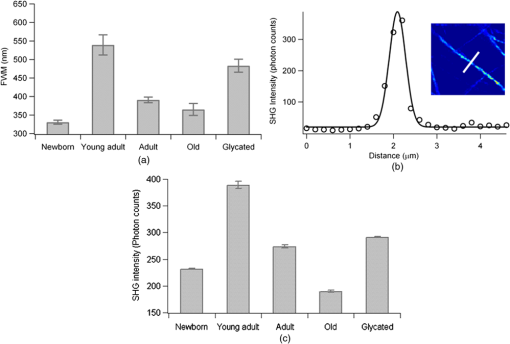 For each age condition, the analysis of PSHG signals was performed on images taken in different regions of the whole sample surface. Only pixels exhibiting a total SHG signal above are considered, which ensures that the measurements are not dominated by noise.16 Typical retrieved images for the orientation () and molecular order () parameters are shown in Fig. 3(a). is found to be close to 0 in all cases, indicating cylindrical-symmetry distributions along the fibrils.16 The angle is well correlated with the macroscopic fibril orientation. The -image shows a large distribution of values, most probably due to intrinsic heterogeneities in fibril morphologies and to the diversity present within the rat’s population of each age-group. However, in spite of such intra-age heterogeneities, inter-age variations are pronounced and can be clearly observed [Fig. 3(c), ANOVA on subregions], with a significance probability below 5% except between adult and young adult cases. The maximum molecular order (high values) is obtained for the newborn and old collagens. Since molecular order is representative of how fibrils are oriented with respect to each other, a large value indicates a low degree of orientational freedom for molecules constituting the fibrils, also interpreted as a lower aperture of their angular distribution [Fig. 3(d)].9–11,16 The large value observed in the newborn group can therefore be attributed to higher structural constraint and rigidity, which is a priori contradictory with the higher elastic modulus observed in newborn collagen three-dimensional (3-D) matrices.17 Elasticity can be however ascribed to a more macroscopic scale effect related to the length of fibrils, which appears to increase with age. Finally, young adult fibrils show a lower molecular order, signature of a more complex assembly of individual fibrils within their thicker fibers. Fig. 3(a) Images deduced from PSHG analysis in a young adult sample: averaged orientation () of the fibrils relative to the horizontal axis obtained from the circular decomposition angle (see text) and retrieved molecular order () parameter. (b) Typical PSHG data, recorded in the area represented by the white square. (c) Comparison of the averaged values (mean and SEM) for different age-groups and ribose-glycated collagen fibrils ( subregions of the SHG images). The lines indicate significant differences in mean values between conditions (One way ANOVA, Tukey HSD test). (d) Dependence of relative to the angular cone aperture () of molecules inside fibrils assemblies. Schematic representation of the molecular orientational distribution in the collagen fibril (represented by the grey cylinder), showing in polar plots the probability of orientation of individual molecules constituting a fibril along the cylinder axis. 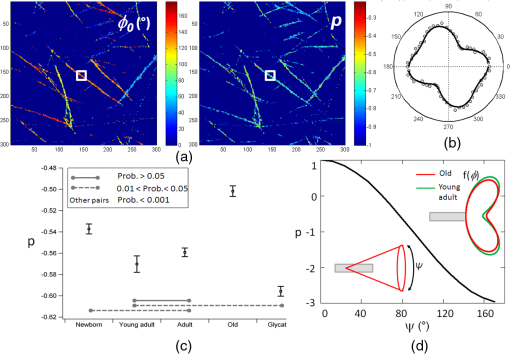 For the glycated collagen, data shown in Figs. 2 and 3 indicate that at a microscopic scale, in vitro glycation does not corroborate with an advanced-age situation, as it has been so far described in the literature.8 Taken together, our data are in agreement with a recent SHG analysis of the collagen index orientation in mice dermal tissue which shows that younger skin exhibits a fine texture whereas aged skin displays a rougher texture.6 In addition, they suggest that in vitro glycated collagen does not represent a good model for studying aged collagen at the microscopic scale. 4.ConclusionWe demonstrate the feasibility of a direct microscopic analysis of different-age type I collagen fibrils using PSHG microscopy. The observed differences between the microscopic structure of newborn and adult fibrils are not necessarily correlated with their macroscopic elasticity properties. This information can be potentially used in tissue samples, particularly in situations where early diagnosis based on subtle morphological changes is a challenge. AcknowledgmentsWe thank Philippe Réfrégier for his contribution in the development of the theoretical model. This work is supported by CNRS, the Région Provence Alpes Côte d’Azur and the Région Champagne-Ardenne. ReferencesN. C. AveryA. J. Bailey,
“The effects of the Maillard reaction on the physical properties and cell interactions of collagen,”
Pathol. Biol., 54
(7), 387
–395
(2006). PTBIAN Google Scholar
K. Mikulikováet al.,
“Study of postranslational non-enzymatic modifications of collagen using capillary electrophoresis/mass spectroscopy and high performance liquid chromatography/mass spectroscopy,”
J. Chromatogr. A, 1155
(2), 125
–133
(2007). http://dx.doi.org/10.1016/j.chroma.2007.01.020 JCRAEY 0021-9673 Google Scholar
R. G. PaulA. J. Bailey,
“Glycation of collagen: the basis of its central role in the late complications of ageing and diabetes,”
Int. J. Biochem. Cell Biol., 28
(10), 1297
–1310
(1996). http://dx.doi.org/10.1016/S1357-2725(96)00079-9 IJBBFU 1357-2725 Google Scholar
G. Kesava Reddy,
“Cross-linking in collagen by nonenzymatic glycation increases the matrix stiffness in rabbit achilles tendon,”
Exp. Diab. Res., 5
(2), 143
–153
(2004). http://dx.doi.org/10.1080/15438600490277860 EDRXBI 1687-5303 Google Scholar
M. Johannes Koehleret al.,
“Morphological skin ageing criteria by multiphoton laser scanning tomography: non-invasive in vivo scoring of the dermal fibre network,”
Exp. Dermatol., 17
(6), 519
–523
(2008). http://dx.doi.org/10.1111/j.1600-0625.2007.00669.x EXDEEY 0906-6705 Google Scholar
S. Wuet al.,
“Quantitative analysis on collagen morphology in aging skin based on multiphoton microscopy,”
J. Biomed. Opt., 16
(4), 040502
(2011). http://dx.doi.org/10.1117/1.3565439 JBOPFO 1083-3668 Google Scholar
M. J. Koehleret al.,
“In vivo assessment of human skin aging by multiphoton laser scanning tomography,”
Opt. Lett., 31
(19), 2879
–2881
(2006). http://dx.doi.org/10.1364/OL.31.002879 OPLEDP 0146-9592 Google Scholar
J-Y. Tsenget al.,
“Multiphoton spectral microscopy for imaging and quantification of tissue glycation,”
Biomed. Opt. Express, 2
(2), 218
–230
(2011). http://dx.doi.org/10.1364/BOE.2.000218 BOEICL 2156-7085 Google Scholar
P. Stolleret al.,
“Polarization-modulated second harmonic generation in collagen,”
Biophys. J., 82
(6), 3330
–3342
(2002). http://dx.doi.org/10.1016/S0006-3495(02)75673-7 BIOJAU 0006-3495 Google Scholar
R. M. WilliamsW. R. ZipfelW. W. Webb,
“Interpreting second-harmonic generation images of collagen I fibrils,”
Biophys. J., 88
(2), 1377
–1386
(2005). http://dx.doi.org/10.1529/biophysj.104.047308 BIOJAU 0006-3495 Google Scholar
S. Psilodimitrakopouloset al.,
“In vivo, pixel-resolution mapping of thick filaments’ orientation in nonfibrilar muscle using polarization-sensitive second harmonic generation microscopy,”
J. Biomed. Opt., 14
(1), 014001
(2009). http://dx.doi.org/10.1117/1.3059627 JBOPFO 1083-3668 Google Scholar
D. Aït-Belkacemet al.,
“Influence of birefringence on polarization resolved nonlinear microscopy and collagen SHG structural imaging,”
Opt. Express, 18
(14), 14859
–14870
(2010). http://dx.doi.org/10.1364/OE.18.014859 OPEXFF 1094-4087 Google Scholar
I. GusachenkoG. LatourM.-C. Schanne-Klein,
“Polarization-resolved second harmonic microscopy in anisotropic thick tissues,”
Opt. Express, 18
(18), 19339
–19352
(2010). http://dx.doi.org/10.1364/OE.18.019339 OPEXFF 1094-4087 Google Scholar
N. Fourreet al.,
“Extracellular matrix proteins protect human HT1080 cells against the antimigratory effect of doxorubicin,”
Cancer Sci., 99
(8), 1699
–1705
(2008). http://dx.doi.org/10.1111/cas.2008.99.issue-8 CSACCM 1347-9032 Google Scholar
G. Saidet al.,
“Impact of carbamylation and glycation of collagen type I on migration of HT1080 human fibrosarcoma cells,”
Int. J. Oncol., 40
(6), 1797
–1804
(2012). IJONES 1019-6439 Google Scholar
J. Duboissetet al.,
“Generic model of the molecular orientational distribution probed by polarization resolved second harmonic generation,”
Phys. Rev. A, 85 043829
(2012). http://dx.doi.org/10.1103/PhysRevA.85.043829 PLRAAN 1050-2947 Google Scholar
S. L. Wilsonet al.,
“The effect of collagen ageing on its structure and cellular behavior,”
Proc. SPIE, 8222 822210
(2012). http://dx.doi.org/10.1117/12.908749 PSISDG 0277-786X Google Scholar
|

