|
|
1.IntroductionNeurogenic inflammation is the inflammatory process whose inductor mechanism is the activation of the nerve fibers. This type of reaction may have a contributing role in the development and aggravation of psoriasis,1–3 atopic dermatitis,3–6 urticaria,1 and perioral dermatitis.7 Neurogenic inflammation has also been linked to the formation of keloids and hypertrophic scars8 and to the early stages of rosacea.9 Neurogenic-mediated inflammation increasingly appears to be a factor in the onset and evolution of various skin diseases,4,5,10–12 and the study of interactions between the nervous system and the skin is of great interest for the scientific world. Neurogenic cutaneous inflammation induced by local administration of capsaicin, the hot ingredient of red pepper, is one of the most used study models. In the skin, capsaicin causes a painful burning-like sensation and a erythematous-edematous inflammatory reaction.13 These effects are primarily induced by its direct action on the transient receptor potential vanilloid subtype 1 (TRPV1) channel14 found in the nerve endings of type C fibers. As a consequence of capsaicin action on nerve fibers, proinflammatory neuropeptides are released, especially substance P (SP) and calcitonin gene-related peptide (CGRP),15,16 which initiate an inflammatory process associated with cutaneous vasodilation, an increase of vascular permeability, plasmatic extravasation, and edema.17–19 Capsaicin-induced neurogenic inflammation, by either topical administration20–25 or local injection,13,26,27 has been used in many research projects on human subjects. Topical administration of capsaicin on skin determines a long-term response, as shown in a study where maximum increase in cutaneous blood flow was achieved after 30 to 45 min from application and was maintained even after 1 h.28 It is an easy, noninvasive method with minimal discomfort for the study subjects. Administration of capsaicin by injection provides good control on the dose of active substance, but it is invasive and creates discomfort for the subjects. In previous studies, methods like infrared thermography,27 laser Doppler flowmetry,20–22,26 or marking and measurement of the erythema area on skin with planimetry13,27 were used to evaluate capsaicin’s effects on cutaneous microvascularization.29 However, none of these methods allowed a histomorphological evaluation of capsaicin’s effects on cutaneous microcirculation, and there is little information in scientific literature regarding this aspect. Reflectance confocal microscopy (RCM) allows a noninvasive investigation of cutaneous tissue at a depth of up to 250 μm with a resolution similar to histological examination. This method allows in vivo study of microscopic structures, including some cellular details, in skin layers and real-time observation of different micromorphological parameters.30–32 It is considered an excellent method to evaluate the capillary vessels situated in the dermoepidermal junction, allowing the observation of blood cell dynamics in capillaries and providing the means to analyze the morphology of capillary ansae in dermal papillae.3334.35.–36 This study analyzes the effect of capsaicin on cutaneous microvascularization in human subjects using in vivo RCM. 2.Materials and Methods2.1.SubjectsA total of 29 healthy subjects (, ), ages 18 to 35 (average age: 22.62 years), were enrolled in the study. All subjects graduated or were enrolled in a higher education program, and they participated in the study on a voluntary basis. All subjects signed a written consent after having been fully informed about the study, the confidentiality criteria, the rights, and the ethics criteria. Those with cardiovascular or respiratory diseases, autoimmune diseases, neoplasia, organ transplant, psychiatric disorders, infectious diseases during the previous month, or allergic reactions to the substances used in the study or their derivatives were excluded from the study, as well as those taking medication that might influence the physiological parameters under study. Pregnant or breast feeding women were also excluded from the study. Study participants were asked to avoid psychoactive substances, alcohol, coffee, tea, energy drinks, beverages containing caffeine, smoking, taking drugs, and strenuous physical effort 24 h before being tested. The subjects included in the study were randomly divided into two groups. A test group with 15 subjects (, ) was administered locally capsaicin dissolved in a vehicle solution (capsaicin treated group—CAP), and a control group with 14 subjects (, ) was administered locally only the vehicle (noncapsaicin treated group – nonCAP). 2.2.Performance of the ExperimentThe study was conducted in the Dermato-oncology Research Laboratory of Carol Davila Medicine and Pharmacy University, Bucharest, after the approval of the local Ethics Committee. The experiments were performed in the afternoon (12 p.m. to 6 p.m.) at a room temperature of and a humidity of . They lasted approximately 2 h for each subject. Following their arrival at the laboratory, the subjects were seated comfortably and given a period of 45 min to adjust to the local conditions. During that time, they filled in the forms of inclusion in the study, and they watched a slide show on the monitor. At the end of the adjustment period, the region to be investigated was delimited on the dorsal side of the nondominant hand. Any previous injuries were avoided, as were any maneuvers that might cause an inflammatory reaction at the investigated region. Cutaneous microvascularization was evaluated using in vivo RCM. Two sessions of measurements were performed for every subject. Initial evaluation allowed the identification of a baseline level for the parameters under study. The second measurement session was performed after the topical administration of the capsaicin solution (for subjects in the CAP group) or the vehicle administration (for subjects in nonCAP group) on the investigated area. Cutaneous microvascularization changes in the investigated area were quantified at 0, 10, 25, and 40 min after the beginning of administration. 2.3.Administration of Substances on the Investigated RegionCapsaicin (M-2028; Sigma Chemical Co, St Louis, MO) was dissolved at a concentration of 1% in the immersion oil (Crodamol STS oil; Croda Inc., Edison, NJ) used in a standard manner for image acquisition with in vivo RCM. A volume of 7.5 µL capsaicin solution in immersion oil was administered with a pipette (Biohit Proline Single-Channel Pipettor, Variable Volume, 0.5 to 10 µL) on the investigated area in the CAP group. The investigated region was isolated from the adjacent skin with a plastic adhesive disc attached to a metallic ring, which was used to fix the image acquisition device. A similar method of administration was used for subjects in the nonCAP group, but the immersion oil did not contain capsaicin. 2.4.Image Acquisition by RCMWe used the VivaScope® 1500 (Lucid Inc, Rochester, NY) to acquire the images taken by RCM. The wavelength of the laser source was 830 nm, which enabled the visualization of structural details in cutaneous microvascularization at the dermoepidermal junction. The magnetic objective of the microscope was placed in contact with the metallic ring fixed to the skin, without applying any pressure. We acquired sets of images from the center of the investigated region with an area of , situated in the dermoepidermal junction, for every evaluation stage. Although previous studies did not show an influence of image acquisition depth on morphological parameters of papillary capillaries,35 in our study, images were acquired at a similar depth for every subject throughout the experiment. 2.5.Cutaneous Microvascularization EvaluationImages taken by RCM were analyzed using the Java-based image processing and analysis program ImageJ 1.45 ( http://rsbweb.nih.gov/ij/), which can be downloaded and used free of charge for scientific research. For every image, we identified the papillae with visible capillary lumina. After marking the capillary lumina margins, the program allowed automatic calculation of micromorfological parameters. The capillary section area, perimeter, and Feret’s diameter (maximum caliper) of capillaries from the dermal papillae were evaluated (see Fig. 1) for a minimum of 14 dermal papillae per subject. Fig. 1With in vivo RCM images, dermal papillae apear as dark round-oval areas corresponding to dermal tissue, surrounded by bright rings representing cells from the basal layer of the epidermis. Capillary ansae in the structure of dermal papillae appear in transversal section as very dark discs, in which some bright elements can sometimes be noticed representing blood cells. High resolution of the images acquired with RCM enables the micromorphological evaluation of papillary vascular ansae using as parameters the area of section, perimeter, and Feret’s diameter of the capillaries. 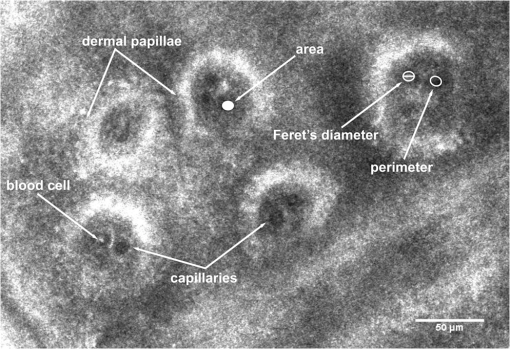 The confocal microscopy images were evaluated in a blinded manner. 2.6.Statistical AnalysisValues of the investigated parameters were calculated for every subject and for every evaluation stage. Values obtained during the time intervals of 0, 10, 25, and 40 min from administration were expressed as a percentage against the baseline value. The program SPSS 12.0 (SPSS, Chicago, IL) was used for statistical analysis of data. Differences between the sexes, and testing of equivalence between the two groups regarding the mean baseline values for each of investigated parameters, were analyzed with one-way between groups analysis of variance (ANOVA). One-way repeated measures ANOVA, followed by Tukey post hoc tests, were used to quantify differences within the two groups for values calculated at every time interval against the baseline for each investigated parameter. When statistically significant differences were revealed, evaluation of correlations existing between these changes was performed, and correlation with the mean baseline values was tested using the Pearson Correlation test. One-way between groups ANOVA was used for comparative analysis between the two groups for the changes calculated against the baseline value for every time interval. The results were presented as an average . A value was considered significant. 3.Results3.1.Mean Baseline ValuesWe performed an overall analysis of mean baseline values for each investigated parameter to evaluate possible differences between female and male subjects, and no statistically significant differences were found (all , ANOVA), as shown in Table 1. Table 1Comparative analysis between female and male subjects of the mean baseline values of the investigated parameters.
Likewise, possible differences in mean baseline values for the investigated parameters between the two groups were analyzed, and no statistically significant differences were revealed (all , ANOVA), as shown in Table 2. Table 2Comparative analysis between the two groups of the mean baseline values of the investigated parameters.
3.2.Changes of Investigated Parameters After Capsaicin or Vehicle Solution ApplicationWe analyzed the values of investigated parameters at 0, 10, 25, and 40 min from administration, searching possible differences between sexes, and similar values were obtained, without revealing statistically significant differences for any parameter. Subsequently, we evaluated within each group the changes of parameters associated to every time interval, and we drew an analysis of correlations between them. The nonCAP group had relatively constant values for capillary section area, perimeter, and Feret’s diameter at 0, 10, 25, and 40 min, without revealing any statistically significant differences (see Fig. 2). Fig. 2Analysis performed within each group regarding the changes of parameters at 0, 10, 25, and 40 min. (a) area of the section, (b) perimeter, and (c) Feret’s diameter of capillaries did not change significantly in the nonCAP group, whereas in the CAP group, a significant elevation was registered 10 min after the application of capsaicin, followed by a slower increasing trend after 25 and 40 min. Error bars represent the standard deviation. *, **, Tukey post hoc test. 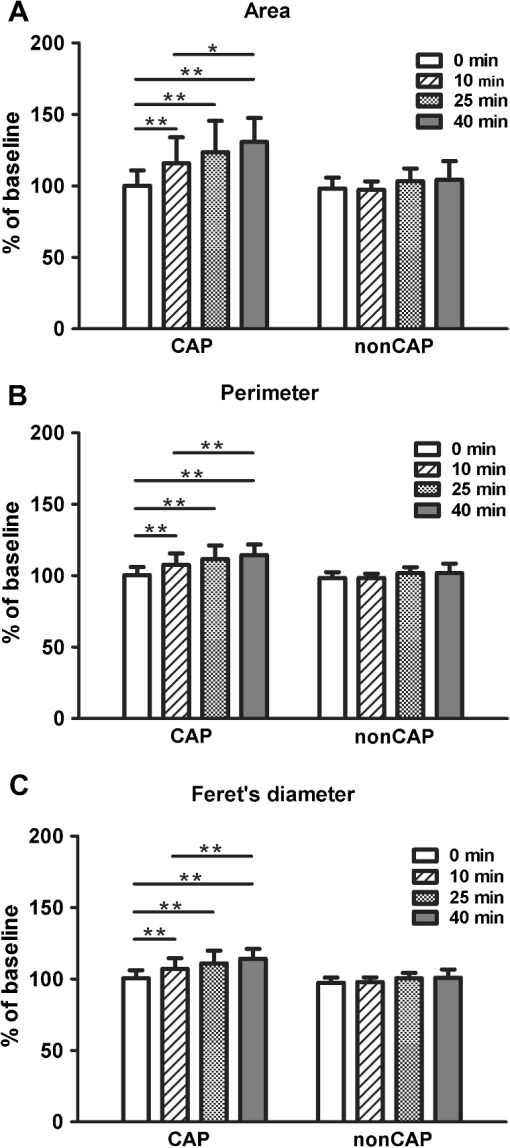 By contrast, the CAP group had a significant increase of these parameters 10 min after capsaicin application, followed by a slower increase trend after 25 min and 40 min, as shown in Figs. 2(a) to 2(c) and 3. This increase was not influenced by baseline values of the above mentioned parameters, since no correlations were revealed between baseline values and changes found at the investigated time intervals (Pearson coefficient between and 0.25327; all ). However, in this group, strong positive correlations were found between values calculated at time intervals of 10, 25, and 40 min after capsaicin application for all parameters (Pearson coefficient between 0.70501 and 0.83267; all ). Subjects with a more intense reaction to capsaicin administration at 10 min maintained a similar response trend at 25 and 40 min after active substance application. These changes in dermal vascular circulation can be easily monitored in real-time examination using RCM (see Videos 1Fig. 2Fig. 3 to 4). Fig. 3Images of RCM of the same dermal papilla acquired (a) 0 min, (b) 10 min, (c) 25 min, and (d) 40 min after the application of capsaicin. Dilation of the lumina of papillary capillaries can be noticed after 10 min. This effect becomes stronger 25 and 40 min after the application of the active substance.  Video 1Papillary dermis vascular circulation at 0 min from capsaicin application (MPEG, 4.75 MB) [URI: http://dx.doi.org/10.1117/1.JBO.17.8.085003.1]. 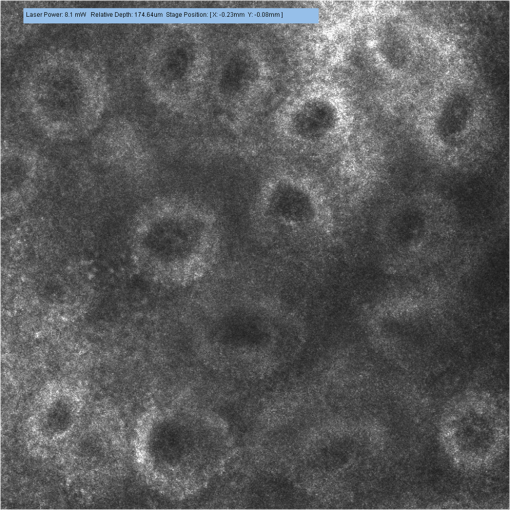 Video 2Papillary dermis vascular circulation 10 min after capsaicin application (MPEG, 5.37 MB) [URI: http://dx.doi.org/10.1117/1.JBO.17.8.085003.2]. 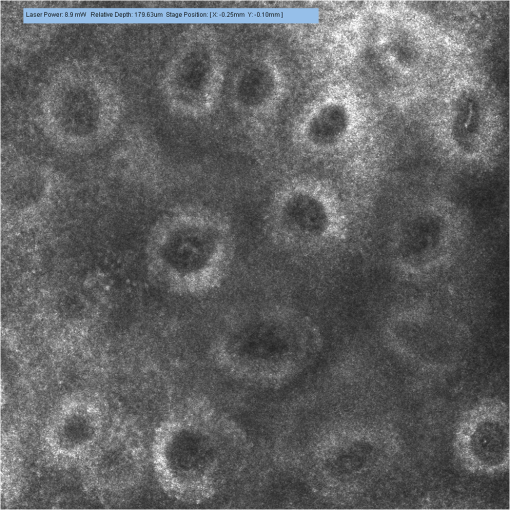 Video 3Papillary dermis vascular circulation 25 min after capsaicin application (MPEG, 5.64 MB) [URI: http://dx.doi.org/10.1117/1.JBO.17.8.085003.3]. 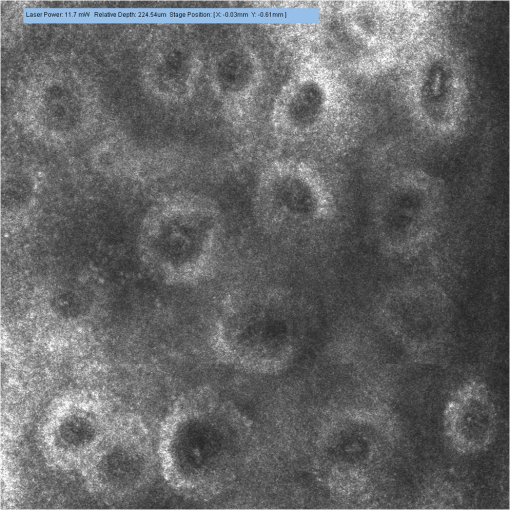 Video 4Papillary dermis vascular circulation 40 min after capsaicin application (MPEG, 5.23 MB) [URI: http://dx.doi.org/10.1117/1.JBO.17.8.085003.4]. 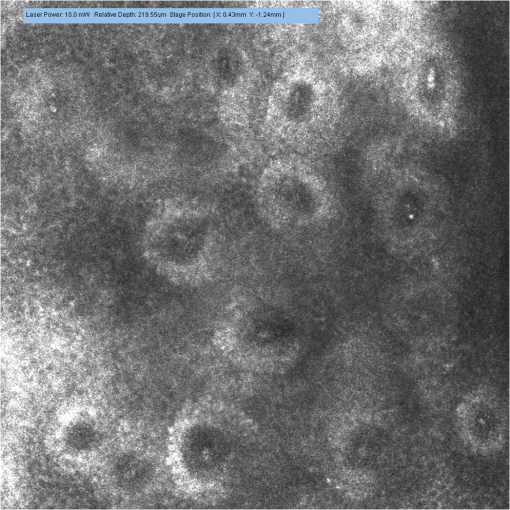 For every time interval, a comparative analysis of values calculated for capillary section area, perimeter, and Feret’s diameter was conducted between the two groups (see Table 3 and Fig. 4). Micromorphological evaluation showed a rapid vasodilation induced by capsaicin. As early as 10 min after active substance application, the capillary section area, perimeter, and Feret’s diameter became significantly larger in the CAP group, and this difference was maintained until the experiment’s conclusion. Table 3Comparative analysis between the two groups of the values at every moment of time of the investigated parameters expressed as a percentage against their baseline value.
Fig. 4Comparative analysis between the two groups of values calculated for the investigated parameters for every time interval. (a) Area of section, (b) perimeter, and (c) Feret’s diameter of capillaries were significantly higher in the CAP group as against the the nonCAP group as early as after 10 min, these differences were mantained after 25 and 40 min. Error bars represent the standard deviation. **, and ***, one way ANOVA test. 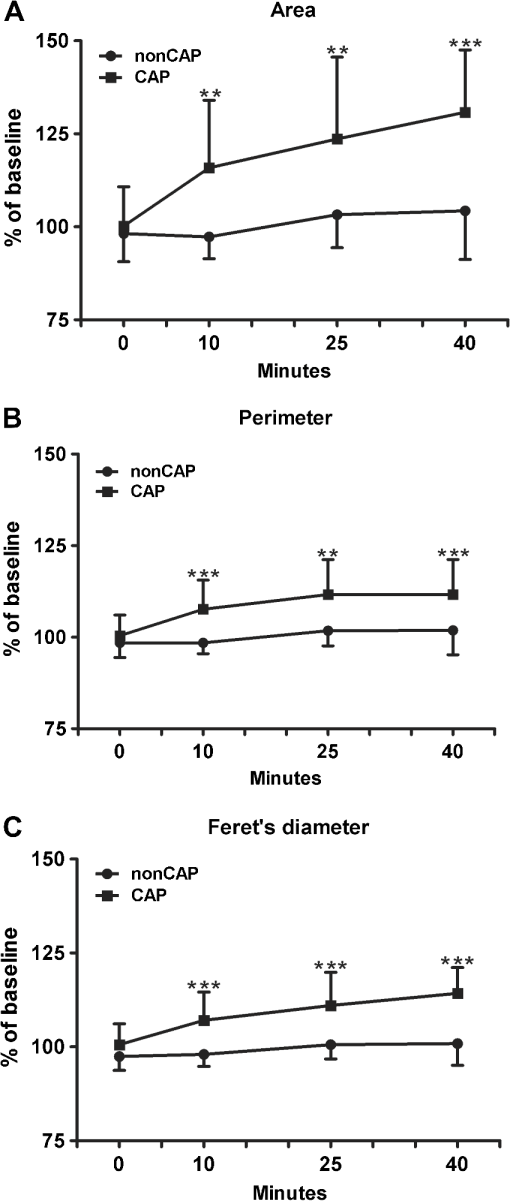 4.Discussions and ConclusionsOur study presents for the first time a micromorphological in vivo evaluation of capsaicin’s effects on cutaneous microcirculation. This study enabled us to report the changes induced by capsaicin at different time intervals compared to baseline values for vascular parameters, and it facilitated the drawing of a time curve of vascular reaction induced by capsaicin. In vivo RCM allows the examination of cutaneous structures to a depth of approximately 250 μm and investigation of vascular reactions in superficial layers of the skin. There are studies suggesting that, in human subjects, neurogenic inflammatory reaction is not homogeneous in superficial and deep dermis, with the vascular response having a distinct aspect in various skin layers.37 This is why in vivo RCM, considered an excellent method of evaluation, particularly for capillary vessels situated at the dermoepidermal junction,35 may constitute a reference technique for future investigation of specific mechanisms through which capsaicin acts in the superior part of cutaneous microvascularization. Administration of capsaicin, dissolved in immersion oil, allows uniform distribution of the active substance on the investigated skin area and strict control of quantity per unit of skin. Moreover, it facilitates image acquisition from the same skin area at each experimental stage. These characteristics may allow the development of a test with clinical applicability, as previous studies revealed that evaluation of neurogenic vascular reaction may constitute a valid test for small-diameter cutaneous nerve fiber functionality.38 The test could be useful for early diagnosis and quantification of therapeutic response in various types of neuropathies affecting thin cutaneous nerve fibers, such as diabetic neuropathy. Investigation of micromorphological aspects of cutaneous neurogenic inflammation using in vivo RCM offers useful information for practitioners and researchers. It could be a potential diagnostic tool for cutaneous inflammatory disorders and could enable identification of new mechanisms by which the nervous system can interfere with physiopathological processes in skin inflammation. AcknowledgmentsResearch funded by grant PN-II-RU-TE-2011-3-0249 from the National University Research Council, Romania. This study is part of the doctoral thesis of Constantin Caruntu that expresses his gratitude for Professor Leon Zagrean from ”Carol Davila” Medicine and Pharmacy University, Bucharest for his guidance and support. Authors wish to thank Dr. Manuela Stanculescu from the Research Institute for Quality of Life, Romanian Academy for statistical analysis and Mihaela Adriana Ghita for her excellent assistance during the course of this work. Special thanks are given to Dr. Monica Neagu from ”Victor Babes” Institute, Bucharest for revising the manuscript. ReferencesS. P. RaychaudhuriS. K. Raychaudhuri,
“Role of NGF and neurogenic inflammation in the pathogenesis of psoriasis,”
Prog. Brain Res., 146 433
–437
(2004). http://dx.doi.org/10.1016/S0079-6123(03)46027-5 PBRRA4 0079-6123 Google Scholar
W. Glinskiet al.,
“Neuropeptides in psoriasis: possible role of beta-endorphin in the pathomechanism of the disease,”
Int. J. Dermatol., 33
(5), 356
–360
(1994). IJDEBB 0011-9059 Google Scholar
T. Scholzenet al.,
“Neuropeptides in the skin: interactions between the neuroendocrine and the skin immune systems,”
Exp. Dermatol., 7
(2–3), 81
–96
(1998). http://dx.doi.org/10.1111/exd.1998.7.issue-2-3 EXDEEY 0906-6705 Google Scholar
C. LiezmannB. KlappE. M. Peters,
“Stress, atopy and allergy: a re-evaluation from a psychoneuroimmunologic persepective,”
Dermatoendocrinol., 3
(1), 37
–40
(2011). http://dx.doi.org/10.4161/derm DERMFJ 1938-1972 Google Scholar
M. Ohshimaet al.,
“Development of mechanisms associated with neurogenic-mediated skin inflammation during the growth of rats,”
Pediatr. Res., 67
(4), 363
–368
(2010). http://dx.doi.org/10.1203/PDR.0b013e3181d026a5 PEREBL 0031-3998 Google Scholar
A. JärvikallioI. T. HarvimaA. Naukkarinen,
“Mast cells, nerves and neuropeptides in atopic dermatitis and nummular eczema,”
Arch. Dermatol. Res., 295
(1), 2
–7
(2003). ADREDL 0340-3696 Google Scholar
F. GuarneriH. Marini,
“An unusual case of perioral dermatitis: possible pathogenic role of neurogenic inflammation,”
J. Eur. Acad. Dermatol. Venereol., 21
(3), 410
–412
(2007). http://dx.doi.org/10.1111/jdv.2007.21.issue-3 JEAVEQ 0926-9959 Google Scholar
S. AkaishiR. OgawaH. Hyakusoku,
“Keloid and hypertrophic scar: neurogenic inflammation hypotheses,”
Med. Hypotheses., 71
(1), 32
–38
(2008). http://dx.doi.org/10.1016/j.mehy.2008.01.032 MEHYDY 0306-9877 Google Scholar
M. Steinhoffet al.,
“Clinical, cellular, and molecular aspects in the pathophysiology of rosacea,”
J. Investig. Dermatol. Symp. Proc., 15
(1), 2
–11
(2011). http://dx.doi.org/10.1038/jidsymp.2011.7 1087-0024 Google Scholar
B. ZegarskaA. LelińskaT. Tyrakowski,
“Clinical and experimental aspects of cutaneous neurogenic inflammation,”
Pharmacol. Rep., 58
(1), 13
–21
(2006). PRHEDU 1734-1140 Google Scholar
C. Căruntuet al.,
“The role of stress in skin diseases,”
Medicina Internă - Internal Medecine., 8
(3), 73
–84
(2011). Google Scholar
C. Căruntuet al.,
“The role of stress in the multifactorial etiopathogenesis of acne,”
Ro. Med. J., 58
(2), 98
–101
(2011). Google Scholar
S. Lutgendorfet al.,
“Effects of relaxation and stress on the capsaicin-induced local inflammatory response,”
Psychosom. Med., 62
(4), 524
–534
(2000). PSMEAP 0033-3174 Google Scholar
M. J. Caterinaet al.,
“The capsaicin receptor: a heat-activated ion channel in the pain pathway,”
Nature, 389
(6653), 816
–824
(1997). http://dx.doi.org/10.1038/39807 NATUAS 0028-0836 Google Scholar
P. Holzer,
“Local effector functions of capsaicin-sensitive sensory nerve endings: involvement of tachykinins, calcitonin gene-related peptide and other neuropeptides,”
Neuroscience, 24
(3), 739
–768
(1988). http://dx.doi.org/10.1016/0306-4522(88)90064-4 NERSD9 0735-2743 Google Scholar
J. D. RichardsonM. R. Vasko,
“Cellular mechanisms of neurogenic inflammation,”
J. Pharmacol. Exp. Ther., 302
(3), 839
–845
(2002). http://dx.doi.org/10.1124/jpet.102.032797 JPETAB 0022-3565 Google Scholar
D. Roostermanet al.,
“Neuronal control of skin function: the skin as a neuroimmunoendocrine organ,”
Physiol. Rev., 86
(4), 1309
–1379
(2006). http://dx.doi.org/10.1152/physrev.00026.2005 PHREA7 0031-9333 Google Scholar
B. VeronesiM. Oortgiesen,
“The TRPV1 receptor: target of toxicants and therapeutics,”
Toxicol Sci., 89
(1), 1
–3
(2006). http://dx.doi.org/10.1093/toxsci/kfj034 TOSCF2 1096-0929 Google Scholar
A. SzallasiP. M. Blumberg,
“Vanilloid (Capsaicin) receptors and mechanisms,”
Pharmacol Rev., 51
(2), 159
–212
(1999). PAREAQ 0031-6997 Google Scholar
S. R. Sinclairet al.,
“Inhibition of capsaicin-induced increase in dermal blood flow by the oral CGRP receptor antagonist, telcagepant (MK-0974),”
Br. J. Clin. Pharmacol., 69
(1), 15
–22
(2010). http://dx.doi.org/10.1111/bcp.2010.69.issue-1 BCPHBM 0306-5251 Google Scholar
H. Wanget al.,
“Ethnic differences in pain, itch and thermal detection in response to topical capsaicin: African Americans display a notably limited hyperalgesia and neurogenic inflammation,”
Br. J. Dermatol., 162
(5), 1023
–1029
(2010). http://dx.doi.org/10.1111/bjd.2010.162.issue-5 BJDEAZ 0007-0963 Google Scholar
A. J. TerkelsenF. W. BachT. S. Jensen,
“Experimental forearm immobilization in humans reduces capsaicin-induced pain and flare,”
Brain Res., 1263 43
–49
(2009). http://dx.doi.org/10.1016/j.brainres.2009.01.054 BRREAP 1385-299X Google Scholar
S. A. Boudreauet al.,
“Vascular and psychophysical effects of topical capsaicin application to orofacial tissues,”
J. Orofac. Pain., 23
(3), 253
–264
(2009). JOROEO 1064-6655 Google Scholar
D. P. Stephenset al.,
“The influence of topical capsaicin on the local thermal control of skin blood flow in humans,”
Am. J. Physiol. Regul. Integr. Comp. Physiol., 281
(3), R894
–R901
(2001). 0363-6119 Google Scholar
P. MohammadianO. K. AndersenL. Arendt-Nielsen,
“Correlation between local vascular and sensory changes following tissue inflammation induced by repetitive application of topical capsaicin,”
Brain Res., 792
(1), 1
–9
(1998). http://dx.doi.org/10.1016/S0006-8993(97)01478-9 BRREAP 1385-299X Google Scholar
W. Koppertet al.,
“The effects of intradermal fentanyl and ketamine on capsaicin-induced secondary hyperalgesia and flare reaction,”
Anesth. Analg., 89
(6), 1521
–1527
(1999). 0003-2999 Google Scholar
J. SerraM. CamperoJ. Ochoa,
“Flare and hyperalgesia after intradermal capsaicin injection in human skin,”
J. Neurophysiol., 80
(6), 2801
–2810
(1998). JONEA4 0022-3077 Google Scholar
B.J. Van der Schuerenet al.,
“Reproducibility of the capsaicin-induced dermal blood flow response as assessed by laser Doppler perfusion imaging,”
Br. J. Clin. Pharmacol., 64
(5), 580
–590
(2007). http://dx.doi.org/10.1111/bcp.2007.64.issue-5 BCPHBM 0306-5251 Google Scholar
C. Căruntuet al.,
“Modern techniques for the assessment of local skin blood flow,”
Acta Medica. Marisiensis., 57
(Suppl. 2), 8
(2011). AMMCDJ 2068-3324 Google Scholar
C. Longoet al.,
“Skin aging: in vivo microscopic assessment of epidermal and dermal changes by means of confocal microscopy,”
J. Am. Acad. Dermatol., ,
(2011). Google Scholar
P. Calzavara-Pintonet al.,
“Reflectance confocal microscopy for in vivo skin imaging,”
Photochem. Photobiol., 84
(6), 1421
–1430
(2008). http://dx.doi.org/10.1111/php.2008.84.issue-6 PHCBAP 0031-8655 Google Scholar
A. Diaconeasaet al.,
“The role of confocal microscopy in the dermato-oncology practice,”
J. Med. Life., 4
(1), 63
–74
(2011). Google Scholar
M. A. Altintaset al.,
“Insight in human skin microcirculation using in vivo reflectance-mode confocal laser scanning microscopy,”
J. Digit. Imag., 23
(4), 475
–481
(2010). http://dx.doi.org/10.1007/s10278-009-9219-3 JDIMEW Google Scholar
M. A. Altintaset al.,
“In vivo reflectance-mode confocal microscopy provides insights in human skin microcirculation and histomorphology,”
Comput. Med. Imag. Graph., 33
(7), 532
–536
(2009). http://dx.doi.org/10.1016/j.compmedimag.2009.04.011 CMIGEY 0895-6111 Google Scholar
J. Hegyiet al.,
“Confocal laser-scanning capillaroscopy: a novel approach to the analysis of skin capillaries in vivo,”
Skin Res. Technol., 15
(4), 476
–481
(2009). http://dx.doi.org/10.1111/srt.2009.15.issue-4 0909-752X Google Scholar
T. Yamashitaet al.,
“In vivo assessment of pigmentary and vascular compartments changes in UVA exposed skin by reflectance-mode confocal microscopy,”
Exp. Dermatol., 16
(11), 905
–911
(2007). http://dx.doi.org/10.1111/exd.2007.16.issue-11 EXDEEY 0906-6705 Google Scholar
C. Forsteret al.,
“Neurogenic flare responses are heterogeneous in superficial and deep layers of human skin,”
Neurosci. Lett., 185
(1), 33
–36
(1995). http://dx.doi.org/10.1016/0304-3940(94)11218-8 NELED5 0304-3940 Google Scholar
A. Caselliet al.,
“Validation of the nerve axon reflex for the assessment of small nerve fibre dysfunction,”
J. Neurol. Neurosurg. Psychiatry., 77
(8), 927
–932
(2006). http://dx.doi.org/10.1136/jnnp.2005.069609 JNNPAU 0022-3050 Google Scholar
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

