|
|
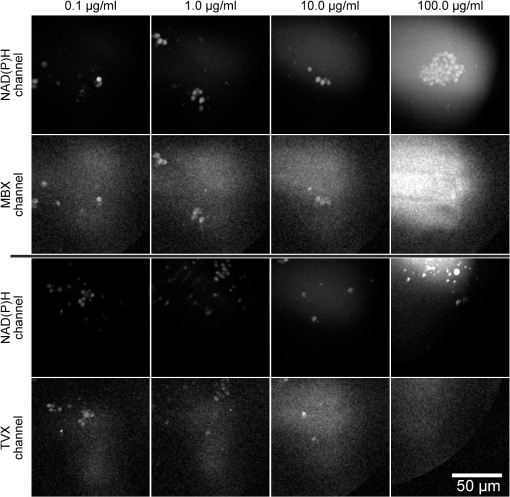
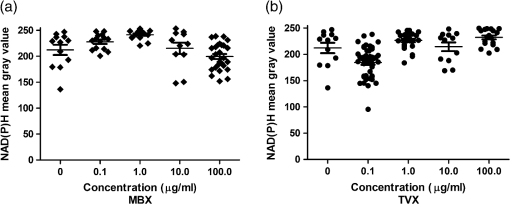
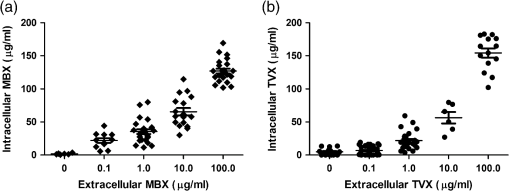
1.IntroductionMultiphoton microscopy (MPM) is a proven technology with applications beyond subjective imaging. Some of the benefits of this unique form of nonlinear imaging include high resolution clinical and veterinary imaging with improved contrast of autofluorescent materials when compared to single-photon confocal microscopy (SPM) or nonconfocal fluorescence microscopy. MPM has the highest resolution of any noninvasive clinical imaging technology. One of the limitations to this approach is the significant cost of equipment, upkeep and training time. These limitations are fading away with improvements in solid state lasers and increased focus on user interfaces. The key difference between single photon microscopy and MPM is that MPM utilizes femtosecond pulsed laser light to excite fluorescent molecules. There are a wide range of applications for MPM that are based on autofluorescence [biological molecules like NAD(P)H, FAD and certain drugs], second harmonic generation (e.g., collagen), and multiphoton-enhanced photoluminescence (e.g., metal nanoparticles). For recent reviews, see Konig et al.1 and Prow et al. 2 Sheep are of great economic value and considered very important animal food sources, particularly in countries where certain diseases that may require an antibiotic treatment are endemic. Some of these diseases are caused by intracellular bacteria such as Salmonellosis, Brucellosis, and Campylobacteriosis. Penetration of antibiotics into various host cells, especially immune cells, is very important to their effectiveness against various microbes, particularly intracellular bacteria. Penicillins, cephalosporins, and aminoglycosides enter cells very poorly and for that reason, they are inactive against intracellular bacteria although they are highly effective antibiotics.3 In order to be active intracellularly, an antibiotic needs to be in direct contact with bacteria which mostly exist in the phagosome of the phagocytic cell.4 We foresee MPM being used more broadly as a noninvasive means to evaluate the presence or absence of autofluorescent molecules of interest in a variety of settings. One area that has yet to be explored is the analysis of autofluorescent antibiotics. Our hypothesis was that MPM could be used to quantify the levels of two important antibiotics, trovafloxacin (TVX) and marbofloxacin (MBX). The reported maximal excitation and emission wavelengths for fluoroquinolones vary but are largely agreed to have two excitation bands at 300 to 350 nm and 245 to 290 nm; the emission band is also broad at 440 to .5 We are the first to explore the use of MPM to evaluate the feasibility of using MPM as a tool to examine TVX and MBX concentrations in sheep neutrophils. As a consequence, we have also gained knowledge into the pharmacokinetics of these important antibiotics in living neutrophils. To our knowledge, this is the first report describing the use of MPM to monitor autofluorescent antibiotic levels. The mechanism of quinolone penetration into cells is not well defined. Using a radioisotopic technique in human neutrophiles, Vazifeh et al. postulated that intracellular accumulation of levofloxacin occurred by a solely passive process.6 On the other hand, Mémin et al. reported that pefloxacin was actively transported at 42°C in in vitro study.7 It may be that quinolones can penetrate into cells passively in normal physical conditions, while some may be actively transported when the temperature or pH is modified.7 TVX is able to concentrate inside phagocytic and nonphagocytic cells, attaining concentrations numerous times greater than those seen extracellularly. A study using radiolabeled TVX, with human polymorphonuclear leukocytes (PMNs) and human peritoneal macrophages, demonstrated that TVX at therapeutic extracellular concentrations reached intracellular concentrations in PMNs of 10-fold or more greater than extracellular levels.8 These rates were a little higher than those obtained with ofloxacin, lomefloxacin, and sparfloxacin, possibly because the greater hydrophobicity of TVX allowed it to cross the cell membrane more easily. Interestingly, TVX remained intracellularly active in PMNs, and its uptake by the same cells was markedly augmented at 4°C regardless of cell viability. On the other hand, van den Broek et al. found that TVX exhibited concentration-independent activity against Staphylococcus aureus that was lower in human granulocytes or monocytes than in a cell-free medium.9 However, Facinelli et al. confirmed that TVX was highly active against all tested Listeria monocytogenes strains and was more intracellularly active than sparfloxacin, ciprofloxacin, and ofloxacin.10 There is limited information about the infiltration of veterinary quinolones into cells. One report stated that enrofloxacin was accumulated in canine white blood cells (WBCs) and alveolar macrophages (AMs) to concentrations greater than 10 times the minimum inhibitory concentration (MIC) for many susceptible pathogens.11 Another study found that MBX reached a concentration in the AMs greater than those seen in plasma and epithelial lining fluid after oral administration in dogs.12 MBX accumulated in AMs at concentrations higher than the mean MICs of major bacterial diseases in dogs, making it suitable for a treatment of intracellular pathogens or infectious diseases associated with pulmonary macrophage penetration. Previous studies reported that TVX and MBX are autofluorescence antibiotics.13,14 In this study, we investigated, for the first time, the penetration of TVX and MBX into isolated sheep neutrophils using MPM. 2.Materials and Methods2.1.Antibiotics and Sheep Blood SamplesAll procedures were approved by the Production and Companion Animal Ethics committee at UQ (PAH/MTCOTT/381/09). MBX and TVX as trovafloxacinmesylate were kindly supplied as a pure material by the Pfizer Inc. (Eastern Point Road, Groton, Connecticut 06340). Aqueous solutions of each drug were prepared initially and adjusted to required concentrations. Sheep were held at Mount Cotton Farm (Karreman Group, Mount Cotton, Queensland, Australia). Blood samples were collected from six sheep (10 ml each) in 5 ml lithium heparin tubes (Vacuette, Interpath Services Pty Ltd, Heidelberg West, VIC Australia, Greiner Bio One, Kremsegg, Austria), using 20G needles and VacutainersHolde (Becton Dickinson Pty. Ltd., North Ryde, NSW Australia). Blood was transferred at room temperature into the lab for neutrophil isolation. 2.1.1.Neutrophil isolationNeutrophil isolation was performed following a modified procedure from previously reported protocols.15,16 Ten milliliters of blood was diluted with an equal volume of 0.9% NaCl at room temperature. Twenty milliliters of diluted blood was slowly and carefully overlayed onto 5 ml of Ficoll-Hypaque (GE Healthcare, Sydney, NSW Australia) gradient separation media with the pipette tip close to the surface of the media to avoid mixing the blood and the media. The blood was then centrifuged at 500 relative centrifugal force (RCF) for 35 min at 20°C. The top three layers (plasma, monocytes, and isolation media) were carefully removed and disposed of. Neutrophils and all of the isolation media beneath the neutrophils were then carefully aspirated and placed into clean centrifuge tubes. The neutrophil solution was diluted with an equal volume of RPMI media (RPMI 1640, PAA laboratories GmbH, Austria) and inverted few times to suspend the cells. The solution was then centrifuged at 350 RCF for 10 min to obtain the red pellet at the bottom of the tube containing neutrophils and residual red blood cells (RBCs). The supernatant solution was removed carefully with a pipette. To lyse the residual RBCs, 10 ml of sterile water was added into each 10 ml volume of solution and mixed by gently inverting the tubes for 20 s at room temperature. Immediately, 10 ml of 9% NaCl solution were added and mixed well by gently inverting the tubes. The tubes were then centrifuged at 250 RCF for 5 min and the supernatant solution was aspired and discarded. The RPMI media (1 ml) was added to the centrifuged solution and vortexed to resuspend the pellet and then the mixture was diluted with 10 ml RPMI media. The centrifugation step was repeated and 0.25 ml of RPMI was added. The pellet was resuspended and neutrophil count was adjusted to . 2.2.Incubation of Neutrophils with AntibioticsIn this study, neutrophils were incubated in RPMI containing different concentrations of either TVX or MBX (0, 0.1, 1, 10, and ) at 37°C for 30 min as previously suggested.8 After incubation of neutrophils with the drugs, samples were processed for MPM imaging. 2.3.Multiphoton MicroscopyThe neutrophil containing solution (15 μl) was placed on a slide with a coverslip. Four slides were prepared from each concentration and two images were taken per each slide. All multiphoton images were obtained on a DermaInspect system (JenLab GmbH, Jena, Germany) with an ultrashort pulsed modelocked, 80 MHz, Titanium Sapphire MaiTai laser (Spectra Physics, Mountain View, California), which has a tuning range of 710 to 920 nm and an 85 fs pulse width. The tomography system has an integrated TCSPC 830 detector to enable FLIM measurements. A Zeiss Plan-Neoflour oil-immersion objective was used. The excitation wavelength was 740 nm for both compounds, and the average incident optical power was 21 mW at the rear of the objective. Images were taken with a brightness of 540 and a contrast of 950. A band pass filter (Schott BG39) was used to filter the light ahead of the FLIM detectors. This detector contained an array of four fast-response time-correlated single-photon counting detectors to capture single-photon events for later image exporting with SPCImage software. 2.4.Single-Photon MicroscopyThe antibiotic-containing solutions were placed on a slide and imaged with a single-photon Zeiss META 510 confocal microscope. A 405-nm laser was used to excite the sample at 10% power using a 0.3 NA EC Plan-Neofluar objective. This wavelength was chosen as the closest comparison to the 740-nm pulsed excitation source described in the MPM method above. A 508- to 625-nm band pass filter was used to collect similar wavelength light as described in the MPM parameters. 2.5.Analysis of Gray Scale Tiff ImagesAll images were analyzed using imagej 1.43 software to obtain mean gray values (MGV). Tiff images of channels 1 [350 to 450 nm, NAD(P)H] and 3 (515 to 620 nm, quinolones) were exported. The NAD(P)H signal was used to identify a neutrophil and create an outline of that cell. This same outline was then applied to the antibiotic channel for cellular measurements. Twenty neutrophils on average were detected for each concentration from either MBX or TVX solutions using MPM. In the SPM standard curve experiment, five areas () were analyzed per concentration group. The extracellular concentration measurement was made in a location without neutrophils, with a uniform area of interest using only MPM. 2.6.-Plane Perpendicular to the Optical AxesIn order to assure that the images were taken in the center of the -plane perpendicular to the optical axes where the MBX or TVX water solution was contained, we used 10.2 μm fluorescence Nile Red beads (Spherotech Inc. Lake Forest, IL). The beads were present in the same -plane as the MBX or TVX solutions validating the imagining depth. The resulting images were processed as described above, except the beads were excluded from the regions of interest analyzed. 2.7.Statistical AnalysisAll data were analyzed and graphed in GraphPad Prism 5.0.1 (GraphPad Prism Software Inc.) or Microsoft Excel. The significance of variance within an entire group was determined by one-way analysis of variance (ANOVA) and between groups by two-way ANOVA.17 The statistical significance was at . 3.Results3.1.Standard Curve GenerationSolutions of MBX and TVX were prepared in sterile water at 0.0, 0.1, 1.0, 10.0, and . A 2-mm o-ring was used to contain the solutions below the coverslip while imaging and the solutions were prepared with fluorescent beads to assure that the correct -plane was imaged for the standard curve. Figure 1 shows the image analysis results for the standard curve generation using either SPM or MPM. The resulting standard curve ranges were linear for both MBX and TVX with of 0.928 and 0.946 when fit with a linear equation, respectively, using MPM but not SPM. The slopes of the fit lines showed that MBX [] were slightly more suitable for MPM quantification within the range chosen than TVX []. Nevertheless, both had positive slopes and linear profiles within the chosen range of 0.1 to . Fig. 1Marbofloxacin (MBX) and trovafloxacin (TVX) solution-only standard curve raw data and linear regression line fit using multiphoton microscopy (MPM) (closed symbols), but not single-photon microscopy (SPM) (open symbols). MBX is shown in filled diamonds and TVX in closed circles. Panel c shows the linear regression line fits for both MBX (, ) and TVX (, ). The error bars show standard error. 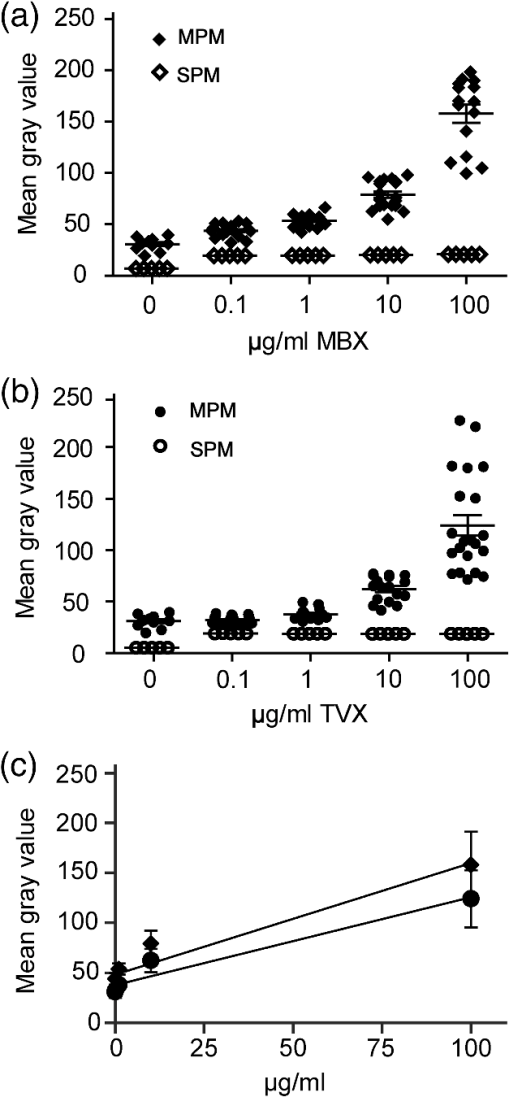 3.2.Neutrophil ImagingSolutions containing freshly isolated neutrophils were treated for 30 min with MBX and TBX at concentrations between 0.0 and . MPM images were taken with emission filters at 350 to 450 nm and 510 to 615 nm for NAD(P)H and antibiotics, respectively (Fig. 2). Neutrophils were sparse or in small aggregates that were easily visible by NAD(P)H imaging. The levels of NAD(P)H remained fairly constant indicating that the cells were metabolically viable and that the treatment regimens did not induce cell death18 (Figs. 2 and 3). In Fig. 2, the two highest concentrations of antibiotics show signal bleed into the NAD(P)H channel. This is a common problem with high levels of fluorescent molecules and introduces an overestimation error in the NAD(P)H measurements in these groups. This is an inherent limitation in some fluorescence-based assays. No blebs or other signs of necrosis were visible. Together, these data indicate that the isolation, treatment, and imaging did not cause overt cytotoxicity. 3.3.Intra- and Extracellular Antibiotic LevelsImage analysis revealed that both antibiotics were more concentrated inside neutrophils than the extracellular media. Figure 4(a) and 4(b) shows the intracellular and extracellular mean gray values for MBX and TVX over the range of concentrations examined. It is apparent from the data shown in Fig. 4 that MBX and TVX are internalized at different rates, which are directly relevant for kinetics studies. 4.DiscussionMPM provides a nondestructive means of analysis for molecules or materials with fluorescent properties in complex media.2 Noninvasive antibiotic quantification in vivo would be valuable for livestock. That said, bringing a multiphoton microscope to a farm may be practically challenging. However, one can hypothesize that a small instrument with a solid state pulsed laser and photodiode detectors could be developed for such a purpose if there were a supporting need. This report describes a feasibility study designed to evaluate the potential for MPM-based antibiotic analysis in WBCs. We carried out a direct measurement of the media fluorescence versus the intracellular fluorescence to gauge the propensity for the cells to uptake TVX and MBX. We found that MBX and TVX treatments were associated with increased levels of intracellular fluorescence above extracellular levels, in a dose-dependent manner. These data support the hypothesis that MPM may be useful for the estimation of fluorescent quinolone levels in neutrophils. However, additional studies need to be done to confirm this hypothesis. In 1999, Vazifeh et al. investigated the uptake of levofloxacin into human neutrophils. Cells were incubated with levofloxacin at 42°C for 5 to 180 min. Our experiments differed in that we incubated at 37°C and for only 30 min. Both studies evaluated the same concentration. Vazifeh et al. found that levofloxacin treatment resulted in a cellular concentration/extracellular concentration ratio of 4 to 6. This is in contrast to our observation. With the same treatment concentration (i.e., ), we found the ratios of 38 and 29. There are several reasons why these differences exist. The model systems and drugs are different but not so divergent as to explain these differences. The major differences are the treatment time and, of course, the methodologies. The levofloxacin data were gathered at 5-min treatment, whereas our TVX and MBX treatment time was 30 min. Further, Vazifeh et al. showed a time-dependent increase in the ratio but did not report values at 30 min. We suggest that all of these variables contribute to these differences and that the methodology is likely the most significant factor. Vazifeh et al. utilized radiolabeled levofloxacin and we used unmodified TVX and MBX. The radiolabeled approach is likely to be far more sensitive and accurate when compared to a microscopy-based technique. Hypothetically, cellular changes induced by treatment could contribute to increased levels of intracellular fluorescence signals. There is, however, precedent for TVX to accumulate to higher intracellular levels than levofloxacin. Pascual et al. reported a cellular to intracellular concentration ratio of TVX-treated human neutrophils to be at 30 min with . Again, a radiolabeled drug was used to acquire these results. Fluoroquinolones utilize several different transporter systems to enter cells.7Ofloxacin has been shown to use aminoacid transporters to gain entry into neutrophils, whereas anion transporters are implicated in norfloxacin transport. Unfortunately, the mechanism for TVX and MBX is not known. However, we foresee that the experimentation to unravel this gap in the literature could be aided by MPM analysis. MPM has the potential to enable real-time assessment before, during, and after transport modulator treatment for the collection on kinetic data. In summary, while there are many unknowns regarding MPM assessment for antibiotic levels in live cells or even in vivo, our data show that there is potential for this approach to be further developed into an effective research tool. Our data showed similar trends to that from radiolabeled drug generated studies, however, our data appeared to overestimate the amount of intracellular drug. Further studies are needed to refine this approach and determine if in vivo application is pragmatic. AcknowledgmentsThe authors would like to thank the Dermatology Research Centre for assistance with imaging. ReferencesK. Koniget al.,
“Applications of multiphotontomographs and femtosecond laser nanoprocessing microscopes in drug delivery research,”
Adv. Drug Deliv. Rev., 63
(4–5), 388
–404
(2011). http://dx.doi.org/10.1016/j.addr.2011.03.002 ADDREP 0169-409X Google Scholar
T. W. Prow,
“Multiphoton microscopy applications in nanodermatology,”
Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol., 4
(6), 680
–690
(2012). http://dx.doi.org/10.1002/wnan.1189 Google Scholar
J. J. Pocidalo,
“Use of fluoroquinolones for intracellular pathogens,”
Rev. Infect. Dis., 11 S979
–S984
(1989). http://dx.doi.org/10.1093/clinids/11.Supplement_5.S979 RINDDG 0162-0886 Google Scholar
H. A. Nguyenet al.,
“Factors influencing the intracellular activity of fluoroquinolones: a study using levofloxacin in a Staphylococcus aureus THP-1 monocyte model,”
J. Antimicrob. Chemother., 57
(5), 883
–890
(2006). http://dx.doi.org/10.1093/jac/dkl079 JACHDX 0305-7453 Google Scholar
F. Canada-Canadaet al.,
“Second-order multivariate calibration procedures applied to high-performance liquid chromatography coupled to fast-scanning fluorescence detection for the determination of fluoroquinolones,”
J. Chromatogr. A, 1216
(24), 4868
–4876
(2009). http://dx.doi.org/10.1016/j.chroma.2009.04.033 JOCRAM 0021-9673 Google Scholar
D. VazifehA. BryskierM.-T. Labro,
“Mechanism underlying levofloxacin uptake by human polymorphonuclearneutrophils,”
Antimicrob. Agents Chemother., 43
(2), 246
–252
(1999). AMACCQ 0066-4804 Google Scholar
E. MeminG. PanteixA. Revol,
“Carrier-mediated system for pefloxacin uptake in human monocytes,”
J. Antimicrob. Chemother., 40
(2), 263
–268
(1997). http://dx.doi.org/10.1093/jac/40.2.263 JACHDX 0305-7453 Google Scholar
A. Pascualet al.,
“Uptake and intracellular activity of trovafloxacin in human phagocytes and tissue-cultured epithelial cells,”
Antimicrob. Agents Chemother., 41
(2), 274
–277
(1997). AMACCQ 0066-4804 Google Scholar
P. J. van den Broeket al.,
“Intracellular activity of trovafloxacin against Staphylococcus aureus,”
J. Antimicrob. Chemother., 44
(2), 193
–199
(1999). http://dx.doi.org/10.1093/jac/44.2.193 JACHDX 0305-7453 Google Scholar
B. Facinelliet al.,
“In vitro extracellular and intracellular activity of two newer and two earlier fluoroquinolones against Listeria monocytogenes,”
Eur. J. Clin. Microbiol. Infect. Dis., 16
(11), 827
–833
(1997). EJCDEU Google Scholar
Hawkinset al.,
“Concentration of enrofloxacin and its active metabolite in alveolar macrophages and pulmonary epithelial lining fluid of dogs,”
J. Vet. Pharmacol. Ther., 21
(1), 18
–23
(1998). http://dx.doi.org/10.1046/j.1365-2885.1998.00103.x JVPTD9 1365-2885 Google Scholar
H. W. Bootheet al.,
“Evaluation of the concentration of marbofloxacin in alveolar macrophages and pulmonary epithelial lining fluid after administration in dogs,”
Am. J. Vet. Res., 66
(10), 1770
–1774
(2005). http://dx.doi.org/10.2460/ajvr.2005.66.1770 AJVRAH 0002-9645 Google Scholar
J. L. VilchezJ. TaoufikiA. Navalon,
“Determination of the antibacterial drug trovafloxacin by solid-phase spectrofluorimetry,”
Anal. Lett., 35
(2), 257
–268
(2002). http://dx.doi.org/10.1081/AL-120002528 ANALBP 0003-2719 Google Scholar
M. A. Garciaet al.,
“Determination of marbofloxacin in plasma samples by high-performance liquid chromatography using fluorescence detection,”
J. Chromatogr. B Biomed. Sci. Appl., 729
(1–2), 157
–161
(1999). http://dx.doi.org/10.1016/S0378-4347(99)00144-9 JCBBEP 0378-4347 Google Scholar
H. OhB. SianoS. Diamond,
“Neutrophil isolation protocol,”
J. Vis. Exp., 17 e745
(2008). Google Scholar
D. W. Siemsenet al., Neutrophil Isolation From Nonhuman Species Neutrophil Methods and Protocols, 21
–34 Humana Press, Totowa, NJ
(2007). Google Scholar
D. S. MooreG. P. McCabeB. A. Craig, Introduction to the Practice of Statistics, W.H. Freeman, New York
(2009). Google Scholar
W. Y. Sanchezet al.,
“Analysis of the metabolic deterioration of ex vivo skin from ischemic necrosis through the imaging of intracellular NAD(P)H by multiphoton tomography and fluorescence lifetime imaging microscopy,”
J. Biomed. Opt., 15
(4), 046008
–046008
(2010). http://dx.doi.org/10.1117/1.3466580 JBOPFO 1083-3668 Google Scholar
|