|
|
1.IntroductionUnderstanding how human tissues face hypoxemic stress over the first hours of hypoxic exposure (HE) is of prime importance for people traveling or working at high altitude as well as for clinical purposes including the management of hypoxemic patients and novel hypoxic preconditioning strategies.1,2 HE-induced change in cerebral tissue oxygenation within this time frame is critical since it coincides with the report of frequent clinical events during mild hypoxic commercial flights (e.g., dizziness, fainting), with the deterioration of cognitive and motor performances in workers or sportsmen at altitude,3,4 as well as with the earliest symptoms of acute mountain sickness (AMS).5,6 All these manifestations are supposed to be linked to perturbations of the cerebral hemodynamics and tissue homeostasis.4 Previous investigations mainly focused on cardiorespiratory and cerebrovascular adaptations to HE characterized by the level of arterial oxygen saturation (), and only few studies monitored hourly changes7,8 in cerebral blood flow (CBF) assessed by transcranial Doppler during HE at rest. Although they suggested that HE may increase the risk of impaired oxygen supply to the brain, Nishimura et al.7 failed to observe significant differences between hypoxic and control normoxic conditions in middle cerebral artery blood velocity (MCAv) measured during 5 h at rest. One reason why Nishimura and coauthors may not have seen differences in MCAv could relate to the use of a mild level of hypoxia (, , ). The cerebrovasculature is exquisitely sensitive to partial pressure of carbon dioxide in arterial blood (i.e., , hypocapnic vasoconstrictive effect) and to a lesser degree to the partial pressure of oxygen in arterial blood (i.e., , hypoxic vasodilatory effect).9 Depending on the prevailing (Ref. 10), a drop in below a certain threshold (40 to 50 mmHg) is required for significant cerebral vasodilation.11 For instance, Ainslie and Poulin10 demonstrated that isocapnic and hypercapnic hypoxia (, ) caused a 24 and 34% increase in CBF compared with changes from normoxic baseline, respectively. In poikilocapnia, a high acute ventilatory response blocked much of the hypoxic response so that the vasodilatory effects of hypoxia were balanced by the vasoconstrictive effects of hypocapnia, resulting in little change in the CBF. Transcranial Doppler (TCD) has been used extensively to assess relative changes in CBF velocity at high altitudes.12,13 TCD can provide quantitative information on cerebral hemodynamic changes at the macrovascular level (i.e., cerebral arteries) but is unable to assess directly the qualitative repercussions of such changes for the tissue at the microvascular level. How much reductions in translate into changes in cerebral but also muscle tissue oxygenation when continuous HE is prolonged over hours remains largely unknown. The impact of hypoxemia on tissue oxygen saturation involves complex mechanisms,14 and both the amplitude and kinetics of tissue deoxygenation when is reduced remain to be clarified.15,16 Animal models have demonstrated, for instance, that changes in oxygen partial pressure of brain tissue may exhibit a significant decoupling pattern compared to experimentally induced swings in (Ref. 17). Tissue-specific sensitivity to short-term hypoxia has been recently emphasized in humans,3,18 but the effect of continuous HE over several hours at rest has not been investigated when regarding skeletal muscle and multiple brain regions. Near-infrared spectroscopy (NIRS) is an optical method that noninvasively monitors regional changes in oxy-, deoxy-, and total hemoglobin by measuring changes in attenuation of NIR light passing through tissue.19 Particularly in the last decade, NIRS has been widely used and recognized as a powerful tool to measure the (mis)balance between oxygen supply and utilization directly in tissue microvessels (venules, arterioles, and capillaries), with a predominant venous contribution (70 to 80%).20,21 Hence, NIRS appears suitable for studying muscle and cerebral hemodynamics and oxygenation responses to challenging environmental conditions as altitude,3,4,18 but has never been used continuously over the early hours of an HE. The aim of the present study was to investigate the kinetics and amplitude of cerebral and skeletal muscle oxygenation responses together with changes in systemic and cardiovascular outcomes over the early 4 h of HE in humans at rest. We hypothesized that tissue oxygenation (especially cerebral) would take longer time to reach a steady-state level compared to systemic (arterial oxygenation) responses. We also hypothesized that brain would show greater deoxygenation responses in the time-course of an exposure to hypoxia sustained for 4 h in resting conditions as compared to skeletal muscle. 2.Materials and Methods2.1.SubjectsEleven healthy male subjects participated in this study. All subjects were nonsmokers and had no history of head injury or cardiorespiratory or neuromuscular diseases. Their average () age, weight, and BMI were years, , and , respectively. The study was approved by the local ethics committee and performed according to the Declaration of Helsinki. All subjects gave their written informed consent to participate in the study and were instructed to drink no alcoholic or caffeinated beverages, and to avoid any physical activity for at least 12 h before each testing day. 2.2.Experimental DesignThe study protocol included two sessions (hypoxic and normoxic sessions) performed at the same time of the day and at least 1 week apart. Subjects seated quietly in a comfortable chair with legs down on the floor, while breathing a gas mixture delivered via a face-mask connected to a mixing gas chamber (Altitrainer 200®, SMTEC, Nyon, Switzerland). After stabilization of the cardiorespiratory variables, baseline data were collected in normoxic condition [inspiratory fraction () of the gas mixture: 0.21] during a 3-min period. Subjects were then exposed rapidly (within 2 min) to 4 h of normobaric hypoxia (, hypoxic session) or normoxia (, normoxic session) and then again to normoxia for 15 min. This time frame may be critical regarding the genesis of hypoxia-induced cerebral perturbations (e.g., AMS symptoms), and the reduced used in the present study (equivalent to an altitude of ) is known to induce values of as frequently observed in clinical practice ( to 90%). Subjects were blinded to the treatment (hypoxic or normoxic session and changes in during each session). Subjects were asked to breathe normally and to refrain from sleeping, talking, or moving to avoid any interference with the normal course of physiological adaptations to the gas inhalation. Temperature and humidity inside the experimental room were maintained at and , respectively. 2.3.Measurement of Systemic Oxygen Saturation/End-TidalSystemic was measured continuously using a pulse oximeter (Pulsox 300, Konica Minolta, Osaka, Japan) placed on the subject’s right index finger, and end-tidal partial pressure () was measured using a respiratory gas monitor (Ohmeda RGM, GE Healthcare, Little Chalfont, UK) connected to the face-mask. 2.4.Measurement of Tissue Oxygenation2.4.1.Device and principleA multichannel NIRS device (Oxymon Mk III, Artinis, The Netherlands) emitting continuous wavelengths of 760- and 850-nm light was used to estimate relative concentration changes (from an initial normoxic baseline of zero) in cerebral and muscle oxygenated-hemoglobin (), deoxygenated-Hb (ΔHHb), and total-Hb (). Theoretical and performance details of NIRS have been previously described.19,22 NIR-determined hemodynamics reflect the dynamic balance between demand and supply in the tissue microcirculation. NIRS has been validated in comparison with washout methods23 and positron emission topography scanning24 and correlates with jugular venous bulb oxygen saturation in healthy volunteers under conditions of hypoxia.25 Because [HHb] is closely associated with changes in venous content and is less sensitive to [tHb] than , [HHb] provides a highly sensitive measure of changes in muscle deoxygenation status due to extraction, while seems to be the most sensitive indicator of regional CBF modifications.26 [tHb] reflects the changes in tissue blood volume within the illuminated area26,27 so that it is used as an index of tissue perfusion.28 In the present study, subjects were instrumented to monitor μMol changes in tissue oxygenation across one muscular and two cerebral sets of probes. 2.4.2.MuscleOxygenation profile was determined over one of the main locomotor muscles to assess potential hypoxia-induced peripheral impairments. One transmitter (Tx)–one receiver (Rx) optode pairs were attached to the skin on the lower third of the belly of the right vastus lateralis (range of 15 to 20 cm above the proximal border of the patella) and in parallel with the long axis of the muscle. Published differential path length of 4.95 cm was used to estimate muscle oxygenation,29 and skinfold measurements were made in the sagittal plane midway between optodes to ensure skin and adipose thickness were below the recommendations to allow the NIR light to properly penetrate through the muscle.30 The distance between the transmitting and receiving optodes was fixed at 4 cm by a plastic probe holder secured to the skin using double-sided tape and covered with a black sweatband maintained with an elastic muff net to shield the optodes from ambient light. 2.4.3.CortexA second set of probes (1 Tx–1 Rx) was placed over the left prefrontal cortex (PFC) to illuminate cortical area between standard Fp1 and F3 locations according to the international EEG 10-20 system.31 PFC synthesizes information from a wide range of brain systems and exerts control over cognitive and executive behavior (e.g., decision-making, movement planning, pacing strategies), so that these associative areas play a central role in the orchestration of thoughts and actions in accordance with internal goals.32,33 The interoptode distance was fixed at 3.5 cm and the probe holder was secured to the skin with double-sided tape and maintained with Velcro headbands. The third set of probes positioned four NIR channels (2 Rx–2 Tx) to span the left premotor and motor cortices (MC) with a specially designed square headset held in place with elastic bandages. Two 3-cm spaced Rx–Tx sets were positioned in the sagittal plane and two 3-cm spaced Rx–Tx sets were positioned in the coronal plane, between standard Cz and C3 locations according to the international EEG 10-20 system.31 These areas have been chosen to reflect absence of brain activation as subjects were asked to remain steady during the protocol. Off-center placement was necessary to illuminate cortical tissue without interference from the sagittal sinus. Subject’s hairs under the optodes were shaved to ensure proper placement (i.e., maximal adherence to the scalp) and maximize signal strength (i.e., strong pulsatile signal). 2.4.4.ProcessingCare was taken to ensure that the attached probes did not constrict the head and did not block scalp blood circulation. Age-related differential path lengths for the brain tissue were used for each subject and each cerebral Rx–Tx set according to previously published tabulated data.34 The four MC NIR pairs were pooled. The positions of the NIR probes were marked on the leg skin with a semipermanent ink marker and precisely redetermined each time over the head to obtain consistent measures over the two testing sessions. Data were recorded continuously at 25 Hz and filtered with a 2-s width moving Gaussian smoothing algorithm before analysis. 2.5.Measurement of Heart Rate Variability and Mean Arterial PressureFull mean arterial blood pressure (MABP) and heart rate variability (HRV) evaluations (i.e., all stages for both sessions) were measured in 7 and 8 out of the 11 subjects, respectively. 2.5.1.Signal acquisitionHeart rate was monitored using the Nexfin HD computer (BMEYE B.V, Amsterdam, Netherlands) by surface four-lead electrocardiography. Continuous estimates of MABP were collected noninvasively with the Nexfin HD monitor, based on the pulsatile unloading of the finger arterial walls using an inflatable finger cuff with a built-in photoelectric plethysmograph. Signals derived from the cuff were converted at a sampling rate of 200 Hz, analyzed, and presented in real time on the Nexfin HD stand-alone device. 2.5.2.Heart rate variability analysisHR and HRV were evaluated over continuous 10-min periods. Cardiac inter-beat (RR) intervals were analyzed (e.g., artifact correction) in accordance with international guidelines.35 The square root of the mean of squared differences between consecutive RR intervals (RMSSD) and the percentage of intervals that vary by from the previous interval (pNN50) were calculated in the time domain. For HRV analysis in the spectral domain, discrete Fourier transform algorithms were applied to RR interval time series and power spectra were quantified with the Kubios software (Biosignal Analysis and Medical Imaging Group, University of Kuopio, Finland). The high-frequency (HF) component of RR ( 0.15 to 0.40 Hz) was used as an index of efferent vagal modulation to the heart (i.e., parasympathetic activity), and the low-frequency (LF) component of RR ( 0.04 to 0.15 Hz) was used to estimate an amalgam of both sympathetic and parasympathetic activity. The low- to high-frequency power ratio () was calculated as an estimate of cardiac sympathovagal balance.36 and are reported here as normalized for total power (0 to 0.40 Hz) minus the very-low-frequency power (0 to 0.04 Hz).35 2.6.Dynamics of Tissue Oxygenation and Cardiorespiratory Responses, , HR, MABP, and NIRS parameters were averaged over 3-min periods at the end of the initial normoxic period (BL), at , 20, 30, 40 and 50 min to study HE-induced adaptations in the first hour, and at BL, , 80, 140, 200, and 240 min and 15 min after return to normoxia () to study HE-induced adaptation over the 4-h exposition period. Steady-state MABP and HRV parameters were collected over 10-min periods at BL, , 80, 140, 200, and 240 min in the 4-h exposition period and at . The same averaging was carried out in normoxic session to evaluate the possible effects of time independent of HE. 2.7.Self-Reported AMS QuestionnairesTo evaluate AMS symptoms, subjects completed self-reported questionnaires before each testing session and in the last 30 min of the 4-h gas exposition period. These evaluations comprised four sections of the Lake Louise AMS questionnaire (LLS, i.e., “headache,” “gastrointestinal distress,” “fatigue/weakness,” “dizzy/light-headedness”),37 the neurologic part of the Environmental Symptom Questionnaire (ESQ-III AMS-C, 11 items graded from 0 to 5 covering “lightheadedness,” “faintness,” “weakness,” “nausea,” “I feel hung-over,” and so on),38 and a visual analog scale for headache (VAS, score from 0 to 100 mm). AMS is defined as an LLS score or an AMS-C score . 2.8.Statistical AnalysisNormality of distribution and homogeneity of variances of the main variables were confirmed using a Skewness-Kurtosis normality test and the Levene’s test, respectively. For each analysis (over the first hour and over the entire gas exposition), two-way () analysis of variance (ANOVA) with repeated measures were performed for each dependent variable. Post hoc Fisher’s LSD-tests were applied to determine a difference between two mean values if the ANOVA revealed a significant main effect or interaction effect. Relationships among changes in tissue oxygenation parameters, cardiorespiratory function parameters, , and symptomatic data were determined by Pearson product correlations. For all statistical analyses, an alpha level of 0.05 was used as the cut-off for significance. Data are presented as in text and tables and as in figures. 3.Results3.1.Adaptations Over the First Hour of HE3.1.1.Arterial oxygenation profileOver the first hour, was significantly reduced during the hypoxic session compared to BL with a plateau from , while no significant changes occurred in the normoxic session (interaction effect, , ) [Fig. 1(a)]. Fig. 1Mean changes () in arterial oxygen saturation () and end-tidal partial pressure () during exposure to 12% hypoxia (solid lines). Dashed lines identify values for the control normoxic session. Data are shown over the first hour [panels (a) and (b)] and over 4 h of hypoxic exposure [panels (c) and (d)]. BL, baseline; , 15 min after normoxic washout. * versus BL; ‡ versus BL and ; $ versus normoxic session. 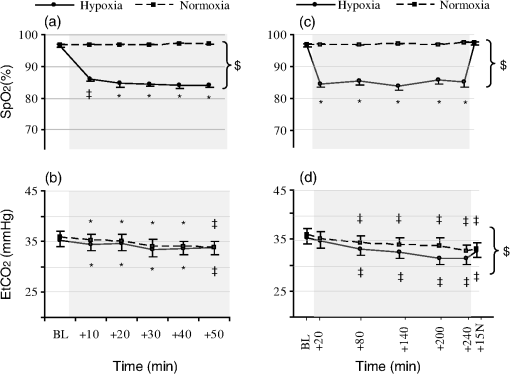 3.1.2.Tissue oxygenation profilesAt the muscle site, the hypoxic session resulted in lower (interaction effect, , ) and higher (interaction effect, , ) from compared with BL, whereas did not change over the first hour of gas exposition in both hypoxic and normoxic sessions [Figs. 2(a) to 2(c)]. The hypoxic session resulted in reduced PFC (interaction effect, , ) and increased (interaction effect, , ) from compared with BL, while increased from in both hypoxic and normoxic sessions (main effect of time, , ) [Figs. 2(d) to 2(f)]. The hypoxic session also resulted in lower MC (interaction effect, , ) and higher (interaction effect, , ) from compared with BL, whereas slightly decreased at in both sessions (main effect of time, , ) [Figs. 2(g) to 2(i)]. As shown in Fig. 2 [especially, Figs. 2(e) and 2(g)], 30 to 40 min was required to stabilize the time course of oxygenation changes for PFC and MC at their highest magnitude. Fig. 2Mean changes () in oxy-([]), deoxy-([HHb]), and total-hemoglobin ([tHb]), during the first hour of exposure to 12% hypoxia (solid lines). Dashed lines identify values for the control normoxic session. Data are shown for the vastus lateralis muscle [panels (a) to (c)], for the prefrontal cortex [PFC, panels (d) to (f)], and for the motor cortex [MC, panels (g) to (i)]. BL, baseline. * versus BL; ‡ versus BL and ; $ versus normoxic session. 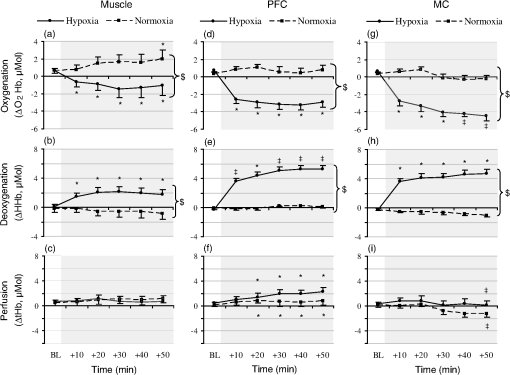 In the hypoxic session, changes in muscle [HHb], [], and [tHb] did not correlate with , whereas PFC and MC exhibited high inverse correlation levels with (at : , and , , respectively). Also, muscle [Hb] changes did not correlate with cerebral [Hb] changes over the first hour of HE. and were tightly correlated between PFC and MC (, and , , respectively), but there was no significant correlation between cerebral sites for (). 3.1.3.Cardiorespiratory function responsesOver the first hour, decreased slightly from compared to BL both in the normoxic and hypoxic sessions ( at , main effect of time, , ) [Fig. 1(b)] but correlated neither to nor to tissue oxygenation changes (). HR remained unchanged through the first hour in the normoxic session, whereas it increased significantly during the hypoxic session ( at , main effect of session, , ). MABP was not notably affected by gas exposition over the first hour, whatever the session. 3.2.Adaptations Over 4-h HE3.2.1.Arterial oxygenation profileDuring the hypoxic session, plateaued at from , while no changes were observed in the normoxic session [Fig. 1(c), interaction effect, , ]. 3.2.2.Tissue oxygenation profilesAt the muscle site, the hypoxic session resulted in decreased from compared to BL [Fig. 3(a), interaction effect, , ]. Increased compared to BL was observed until during the hypoxic session, while a significant decrease in occurred in the normoxic session from [Fig. 3(b), interaction effect, , ]. Significant reduction in muscle was observed at in both sessions [Fig. 3(c), main effect of time, , ]. During the hypoxic session, PFC (interaction effect, , ) and MC (interaction effect, , ) was lower compared to BL, while it increased significantly in the normoxic session from in PFC and in MC [Figs. 3(d) and 3(g)]. The hypoxic session induced a marked increased in compared to BL in both PFC (interaction effect, , ) and MC (interaction effect, , ) [Figs. 3(e) and 3(h)]. Both hypoxic and normoxic sessions resulted in significant cerebral increases [Figs. 3(f) and 3(i)] from in PFC (main effect of time, , ) but only from in MC (main effect of time, , ). Fig. 3Mean changes () in oxy-([]), deoxy-([HHb]), and total-hemoglobin ([tHb]), over a 4-h exposure to 12% hypoxia (plenty lines). Dashed lines identify values for the control normoxic session. Data are shown for the vastus lateralis muscle [panels (a) to (c)], for the PFC [panels (d) to (f)], and for the MC [panels (g) to (i)]. BL, baseline; , 15 min after normoxic washout. * versus BL; ‡ versus BL and ; $ versus normoxic session. 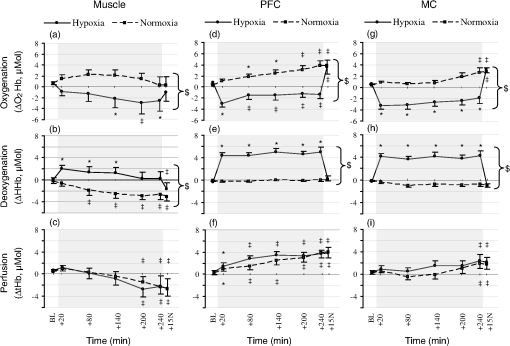 Cerebral hemodynamics assessed by NIRS during the hypoxic session were strongly correlated between PFC and MC (at : , ; , ; , , for ; ; , respectively), with no significant correlations with muscle hemodynamics. At of HE, PFC but not MC was negatively correlated with (, ). As observed during the first hour, muscle hemodynamics did not correlate with over the 4 h of HE. 3.2.3.Cardiorespiratory function and HRV responsesFigure 1(d) shows that was slightly reduced during the hypoxic compared to the normoxic session (main effect of session, , ), even if a progressive hypocapnia was also observed throughout the normoxic session (main effect of time, , ). Increase in MABP appeared significant from in both sessions (main effect of time, , ), this increase being predominant in the normoxic compared to the hypoxic session (Table 1). HR was significantly increased during the hypoxic session (interaction effect, , ) compared to the normoxic session. , MABP, and HR changes at the end of the protocol did not correlate to either or cerebral [Hb] changes (), whatever the session. Table 1Heart rate (HR), mean arterial blood pressure (MABP), and heart rate variability indices over 4 h of gas exposition.
Note: Values are mean±SD over 10-min periods. BL, baseline; +15N, 15 min after normoxic washout; RR, RR intervals; RMSSD, square root of the mean of squared differences between consecutive RR intervals; pNN50, percentage of RR intervals that vary by >50 ms from the previous interval; LF, low-frequency power; HF, high-frequency power; nu, normalized units. Changes in time and frequency domain indices for HRV are shown in Table 1. decrease and increase over time were significantly more pronounced during the hypoxic session compared to the normoxic session (main effect of session, , ). However, the increase in ratio did not differ significantly between sessions. RR intervals and pNN50 were significantly lower throughout the hypoxic session compared to the normoxic session (interaction effects, RR, , ; pNN50, , ), while RMSSD changes did not reach statistical significance, whatever the session. 3.2.4.Self-reported questionnairesLLS increased from to during the hypoxic session (6 out of 11 subjects had scores consistent with AMS), whereas no significant change was observed during the normoxic protocol (interaction effect, , ). AMS-C scores increased from to during the hypoxic session (3 out of 11 subjects had scores consistent with AMS), whereas no significant change was observed during the normoxic session (interaction effect, , ). Headache VAS scores increased from to during the hypoxic session (3 out of 11 subjects had scores ), whereas no significant change was observed during the normoxic session (interaction effect, , ). No significant correlations were found between symptoms and changes in , tissue hemodynamics, or any HRV indices at the end of the hypoxic session (). 4.DiscussionThis is the first study to provide continuous measurements of skeletal muscle and cerebral oxygenation profiles over a 4-h sustained HE in humans. The two major findings were (1) tissue desaturation was significantly delayed compared to the arterial desaturation as deoxygenation pattern induced in the early phase of HE takes 30 to 40 min at the cerebral sites to plateau and (2) a tissue-specific sensitivity to hypoxia was observed, with the magnitude of deoxygenation being more pronounced in the PFC and MC compared to the muscle throughout the 4 h of HE. Finally, the magnitude and kinetics of changes in tissue oxygenation were not associated with those of the cardiorespiratory changes and altitude sickness symptoms appearance observed during HE. 4.1.Decoupling between and Muscle/Cerebral Oxygenation Over a 4-h Sustained HEConcomitant cerebral oxygenation and reduction have been observed in unacclimatized trekkers at moderate altitudes or during progressive ascent to high altitude.39,40 While the magnitude of reduction in the systemic and local tissue oxygenation status has been described during hypoxic exercise loading,41,42 their time courses under prolonged hypoxic resting conditions is poorly understood. In the present study, we observed a significant deoxygenation in muscle and cerebral tissues (PFC and MC) after 10 min of resting HE. While the reduction in () plateaued after 20 min of HE, our findings indicate a time to plateau of 30 to 40 min for PFC and MC and of 10 min for the skeletal muscle. Since cerebral oxygenation is influenced both by and CBF, the difference between cerebral oxygenation and patterns may be explained by the alteration in CBF. In an experimental setting similar to the present one, Nishimura et al.7 observed that blood velocity in the middle cerebral artery (MCAv) was unchanged after 1 h of HE () and did not differ compared to normoxic conditions after 2 h, while was reduced by 2 to 3 mmHg and MABP was unchanged. However, Huang et al.43 provide evidence supporting that the rise in CBF over the first hours of poikilocapnic hypoxic exposure at high altitude is delayed. Using Doppler ultrasound, they found a small but nonsignificant increase in blood velocities in the internal carotid and vertebral arteries after 2 to 4 h at higher altitude (4300 m). Similarly, Teppema et al.8 showed unchanged CBF during 4 h of poikilocapnic hypoxia (). By contrast, 12 to 24 h after arrival at high altitudes (3475 to 4559 m), several TCD studies found increases of 20 to 27% in MCAv.14,44,45 These ultrasound findings are in accordance with previous results using other techniques. Severinghaus and coauthors,46 using the Kety–Schmidt nitrous oxide washout method to analyze changes in CBF, found a 24% increase at 6 to 12 h and a 13% increase at 3 to 5 days after arrival at 3810 m. Hence, the kinetic of CBF changes, particularly in the early hours, in response to prolonged HE may explain, at least in part, the longer time needed to reach stable deoxygenation level within cerebral tissue compared to . It has been suggested that cerebral oxygenation is strongly dependent on arterial partial pressure () due to its effect on CBF.47 It could be postulated that cerebral oxygenation patterns cannot plateau until has reached a given critical or stable value. The time-course of changes in the present study indicates that early modest hypocapnia does not explain the specific cerebral oxygenation profiles observed in response to HE since similar values were observed over the first hour of normoxia and hypoxia. Consistent with previous reports,5,7 prolonged HE induced significant hypocapnia compared to the normoxic session. However, no relationship between and tissue oxygenation was found over time, suggesting that had no specific effect on cerebral oxygenation, even after several hours of HE. The heterogeneity of individual responses to poikilocapnic hypoxia may explain part of the variability in the cerebrovascular tone response, but it is likely that did not strongly influence cerebral (de)oxygenation in our study. It is important to emphasize that NIRS measures the balance between oxygen supply and utilization. NIRS-derived deoxygenation, as observed in our study, refers to a mix between the pre- and postcapillaries state, thus including a decrease in arterial content and also possible mechanisms of exacerbated oxygen extraction to preserve the resting cerebral metabolic rate of oxygen (). There are few field studies of brain oxygen consumption or extraction at altitude because such studies would require arterial and venous (jugular) measurements. Moller and coauthors48 studied and CBF using the Fick and the Kety–Schmidt techniques in nine acclimatized subjects at rest and during exercise at sea level and at 5260 m. Despite profound changes in breathing, no changes were seen in both CBF and oxidative metabolism. In the same way, the was unchanged compared with sea-level values in five subjects investigated within the first 24 h after arrival at 3810 m by Severinghaus and coauthors.46 These data suggest a critical adjustment of the oxygen extraction fraction in response to hypoxemia, especially in the early hours of HE when the decrease in is not always and/or immediately compensated by an increase in the delivery of (through an increase in CBF). This remains however speculative, as human brain models specifying the physiologic control of oxygen delivery at capillary level (other than hemoglobin- and flow-dependent properties of oxygen transport) are scarce.49 The fact that prolonged HE elicits measurable time delay in changes between and tissue (especially cerebral) oxygenation may be of practical importance when drawing protocols aiming at investigating the effect of hypoxic gas inhalation on tissue responses. A hypoxic wash-in period of 30 min could be recommended in order to reach maximal cerebral tissue deoxygenation levels. 4.2.Tissue-Specific Sensitivity to Sustained HEIt has been suggested that oxygen delivery to tissues is tightly matched by immediate cardiac output changes to meet the peripheral demand in normal subjects at rest.50 Some studies40,51 reported long-term adaptations in regional cerebral oxygenation following trekking at high altitudes. Our results show time-dependent responses and distinct kinetics (amplitude and rate) of cerebral and muscle deoxygenation when is reduced over hours. Following HE onset, muscle [HHb] increased to significance after a delay of 10 min and remained constant over the first hour, whereas cerebral [HHb] (PFC and MC) increased progressively to reach a plateau only after 30 to 40 min. Interestingly, cerebral and muscle tissues respond differently to the offset of gas exposure during the first 15 min of normoxic washout: [HHb] returned promptly to baseline values in cerebral tissue, but not in the muscle tissue (see Fig. 3). The literature provides inconsistent results regarding changes in oxygenation status within the skeletal muscle during few minutes of HE, with either no change41,52,53 or a rapid impact on tissue oxygenation state.54 It is likely that these results are mainly explained by a time-dependent relationship, the level of hypoxia, and a high interindividual variability in muscle responses (especially compared to the brain) to acute hypoxia. Our findings over 1-h HE support a larger sensitivity of the cerebral cortex compared to the muscle, even if qualitative signs of skeletal muscle hypoxemia reached significance already after 10 min. Major differences were observed in cerebral and muscle tissue deoxygenation magnitude throughout HE, with a large increase in [HHb] in PFC and MC compared to the vastus lateralis muscle. Previous results have also revealed that the hypoxic cerebrovascular responsiveness has a relatively slow on-response rate.55 The differences observed between the brain and the quadriceps might be due to differences in basal metabolism and in the microvascular architecture and responsiveness of both tissues.53,56 Hence, muscle capillary recruitment may be a tissue-specific strategy to limit the impact of the decreased arterial saturation.57 From - to 4-h HE, changes in muscle [], [HHb], and [tHb] (Fig. 3) may reflect a decrease in local blood flow and/or quadriceps extraction, raising the possibility that blood flow was shifted to other organs and/or other muscle groups (e.g., those involved in the seated position), independent of gas exposure. This seems a plausible explanation since the decline in [HHb] and [tHb] was similar between normoxic and hypoxic sessions (see Sec. 4.4). In the examined brain regions, increase in perfusion ([tHb]) was slightly different in PFC than MC. A marked increase in cerebral blood volume in PFC, as indicated by the higher [tHb] values, was detected as early as after 20 min of HE and remained fairly constant between 2 and 4 h of HE. In contrast, the increased [tHb] occurred only at 240 min HE in MC region. This may be partly explained when considering the regions of the brain associated with integrating sensory input; i.e., the PFC can be considered as an active region even in resting conditions as it is an area of the brain responsible for attention and motivation.33 One possible explanation for this PFC [tHb] elevation at rest might be the effect of being on the seat for such a prolonged time and consequently engaged in (introspective) thoughts. These two patterns were similar in both conditions despite distinct levels of and are therefore independent of mechanisms associated with HE. 4.3.Cardiorespiratory and NIRS Changes and Altitude Sickness Symptoms During Sustained HEHigh-altitude headache has been proposed to be mainly caused by the acute reduction in cerebral regional oxygen saturation in unacclimatized trekkers,39 and acetazolomide decreases AMS symptomatology, at least in part, by helping to maintain cerebral oxygenation at altitudes up to 5700 m.58 Therefore, it is likely that the level of brain deoxygenation plays a key role in the acclimation process. Cerebral oxygenation has been shown to be influenced by sympathetic activity at rest.59 In the present study, stimulation of sympathetic activity as revealed by HRV indices and increased HR (Table 1) during HE was concomitant with a decrease in tissue (cerebral and muscle) oxygenation. The well-known progressive increase in HR with sustained hypoxia occurred during the first 20 min and was followed by a steady state over the 4 h of HE. The observed unchanged MABP during the first hour may then result from a decline in systemic vascular resistance and supports the absence of strong systemic influences on cerebral oxygenation in the early phase () of physiological adjustments to hypoxia. Also, blood pressure did not differ between conditions when HE was prolonged, even if MABP increased slightly over time. This is in agreement with previous studies reporting that acute short-lasting hypoxia ( day) influences only slightly systemic blood pressure in humans.7,60 Our study and others61,62 demonstrated a change in the sympathetic–parasympathetic balance in response to acute HE at rest, with a sympathetic tone increase together with a decrease in parasympathetic tone (i.e., increase and decrease over time, respectively; see Table 1). Previous studies on AMS debated the fact that increased sympathetic activity during high-altitude exposure may be involved in acute hypoxic adaptations63,64 and that parameters like HRV could permit the predication of AMS susceptibility.64 In accordance with Subudhi et al.,5 we found that being 4 h at significantly increases altitude illness symptoms (i.e., headache, LLS, AMS-C score). These results support this time frame to be critical regarding the genesis of hypoxia-induced cerebral perturbations. However, no relationship among HRV indices, AMS symptoms, and NIRS changes was found in the present study. This confirms recent findings suggesting that changes in cardiac autonomic modulations may not be related to AMS development in the early phase of acute normobaric hypoxia.62 4.4.Methodological ConsiderationsOver the brain, the detected NIR light is affected not only by cortical hemodynamic changes but also by changes in optical properties of all other superficial tissue layers between the optode and the gray matter (e.g., scalp, skull, CSF).65,66 However, to minimize the contribution of hemodynamic changes from the skin/scalp layer (e.g., vasomotion/flow motion induced by mechanical skin irritation and/or sympathetic hyperactivation67,68) in the high-probability banana-shaped photon flux paths, we enlarged interoptode distances (up to 3.5 cm for the brain and 4 cm for the muscle) to reach the maximal light path providing a sufficient signal-to-noise ratio of the optical density measurements.19 Controlled ambient temperature and attention given to ensure head NIRS setup to be noncompressive (i.e., no scalp ischemia/hyperaemia) were also important to reduce the weight of the skin layer (i.e., extracranial blood flow in the skin tissue) in perturbing the observed chromophore concentration changes. We are confident that, taken together, the behavior of the present oxygenation changes most probably reflected the brain tissue oxygenation changes. NIRS penetration depth (i.e., approximately half the interoptode distance) limits its sensitivity to the upper 1 cm of the cerebral cortex,69 so that it is important to consider that the measurements obtained are regional and strictly confined to the zone beneath the sensors. We acknowledge that the observed tissue oxygenation cannot be generalized to whole brain or even to cerebral lobes. In order to investigate the kinetic of cerebral hemodynamic responses to 4-h HE, subjects had to be completely relaxed (without sleeping or speaking) in order to avoid inputs from other sources (e.g., anxiety, voluntary hypo- or hyperventilation, etc.), which would influence the measured response. Thanks to the normoxic control session, we identified the fact that staying awake but totally inactive in a sitting position for a long time might have induced some slight effects on parameters expected to remain constant (e.g., muscle and cerebral tHb on Fig. 2). A gradual decrease in cerebral blood velocity (and ) has been previously reported to occur both in normoxic and hypoxic conditions in subjects sitting for 5 h.7 Accordingly, in our study, concomitant muscle hypoperfusion and cerebral hyperperfusion were likely due to the prolonged position rather than to the nature of the inhaled gas over time. Finally, we used a normobaric hypoxic protocol, and whether our results would have been heightened or different if using hypobaric hypoxia is still an open question. 5.ConclusionOxy- and deoxyhemoglobin changes assessed by NIRS allow understanding of qualitative microcirculation hypoxia-induced adaptive mechanisms. The present study showed distinct (i.e., delayed) behavior of local cerebral hemodynamics compared to arterial systemic desaturation profiles in the first hour of HE. When hypoxia was prolonged over hours, continuous NIRS evaluation also emphasized tissue-specific sensitivity, the cerebral cortex being more sensitive to HE than skeletal muscle. Reduced cerebral oxygenation may reflect an imbalance between oxygen supply and extraction, indicating a potential risk for cellular dysfunction, although it does not necessarily indicate tissue damage. The transition to irreversible neurologic damage may depend on both the duration and the severity of the hypoxic stimulus,70 so that further insights are needed, in which NIRS and biomedical optics have to contribute prominently. AcknowledgmentsFinancial support was provided by the French National Research Agency (Grant No. NT09_653348). ReferencesU. DirnaglK. BeckerA. Meisel,
“Preconditioning and tolerance against cerebral ischaemia: from experimental strategies to clinical use,”
Lancet Neurol., 8
(4), 398
–412
(2009). http://dx.doi.org/10.1016/S1474-4422(09)70054-7 LNAEAM 1474-4422 Google Scholar
T. V. Serebrovskayaet al.,
“Intermittent hypoxia: cause of or therapy for systemic hypertension?,”
Exp. Biol. Med. (Maywood), 233
(6), 627
–650
(2008). http://dx.doi.org/10.3181/0710-MR-267 1535-3702 Google Scholar
S. Vergeset al.,
“Invited review: cerebral perturbations during exercise in hypoxia,”
Am. J. Physiol. Regul. Integr. Comp. Physiol., 302
(8), R903
–916
(2012). http://dx.doi.org/10.1152/ajpregu.00555.2011 0363-6119 Google Scholar
M. H. WilsonS. NewmanC. H. Imray,
“The cerebral effects of ascent to high altitudes,”
Lancet Neurol., 8
(2), 175
–191
(2009). http://dx.doi.org/10.1016/S1474-4422(09)70014-6 LNAEAM 1474-4422 Google Scholar
A. W. SubudhiR. B. PaneraiR. C. Roach,
“Effects of hypobaric hypoxia on cerebral autoregulation,”
Stroke, 41
(4), 641
–646
(2010). http://dx.doi.org/10.1161/STROKEAHA.109.574749 SJCCA7 0039-2499 Google Scholar
P. H. HackettR. C. Roach,
“High-altitude illness,”
N. Engl. J. Med., 345
(2), 107
–114
(2001). http://dx.doi.org/10.1056/NEJM200107123450206 NEJMAG 0028-4793 Google Scholar
N. Nishimuraet al.,
“Decreased steady-state cerebral blood flow velocity and altered dynamic cerebral autoregulation during 5-h sustained 15% O2 hypoxia,”
J. Appl. Physiol., 108
(5), 1154
–1161
(2010). http://dx.doi.org/10.1152/japplphysiol.00656.2009 JAPYAA 0021-8987 Google Scholar
L. J. Teppemaet al.,
“Effects of acetazolamide on ventilatory, cerebrovascular, and pulmonary vascular responses to hypoxia,”
Am. J. Respir. Crit. Care Med., 175
(3), 277
–281
(2007). http://dx.doi.org/10.1164/rccm.200608-1199OC AJCMED 1073-449X Google Scholar
P. N. AinslieJ. Duffin,
“Integration of cerebrovascular CO2 reactivity and chemoreflex control of breathing: mechanisms of regulation, measurement, and interpretation,”
Am. J. Physiol. Regul. Integr. Comp. Physiol., 296
(5), R1473
–1495
(2009). http://dx.doi.org/10.1152/ajpregu.91008.2008 0363-6119 Google Scholar
P. N. AinslieM. J. Poulin,
“Ventilatory, cerebrovascular, and cardiovascular interactions in acute hypoxia: regulation by carbon dioxide,”
J. Appl. Physiol. (Bethesda), 97
(1), 149
–159
(2004). EJAPFN 1439-6319 Google Scholar
A. K. Guptaet al.,
“Thresholds for hypoxic cerebral vasodilation in volunteers,”
Anesth. Analg., 85
(4), 817
–820
(1997). AACRAT 0003-2999 Google Scholar
C. B. Wolff,
“Cerebral blood flow and oxygen delivery at high altitude,”
High Alt. Med. Biol., 1
(1), 33
–38
(2000). http://dx.doi.org/10.1089/152702900320667 HAMBB7 1527-0297 Google Scholar
J. V. Brugniauxet al.,
“Cerebrovascular responses to altitude,”
Respir. Physiol. Neurobiol., 158
(2–3), 212
–223
(2007). http://dx.doi.org/10.1016/j.resp.2007.04.008 RPNEAV 1569-9048 Google Scholar
P. N. AinslieS. Ogoh,
“Regulation of cerebral blood flow in mammals during chronic hypoxia: a matter of balance,”
Exp. Physiol., 95
(2), 251
–262
(2010). http://dx.doi.org/10.1113/expphysiol.2008.045575 EXPHEZ 0958-0670 Google Scholar
I. Almendroset al.,
“Tissue oxygenation in brain, muscle, and fat in a rat model of sleep apnea: differential effect of obstructive apneas and intermittent hypoxia,”
Sleep, 34
(8), 1127
–1133
(2011). http://dx.doi.org/10.5665/SLEEP.1176 SLEED6 0161-8105 Google Scholar
C. Reinkeet al.,
“Effects of different acute hypoxic regimens on tissue oxygen profiles and metabolic outcomes,”
J. Appl. Physiol., 111
(3), 881
–890
(2011). http://dx.doi.org/10.1152/japplphysiol.00492.2011 JAPYAA 0021-8987 Google Scholar
I. Almendroset al.,
“Changes in oxygen partial pressure of brain tissue in an animal model of obstructive apnea,”
Respir. Res., 11
(3),
(2010). http://dx.doi.org/10.1186/1465-9921-11-3 RREEBZ 1465-993X Google Scholar
P. N. Ainslieet al.,
“Alterations in cerebral autoregulation and cerebral blood flow velocity during acute hypoxia: rest and exercise,”
Am. J. Physiol. Heart. Circ. Physiol., 292
(2), H976
–983
(2007). http://dx.doi.org/10.1152/ajpheart.00639.2006 0363-6135 Google Scholar
P. Rolfe,
“In vivo near-infrared spectroscopy,”
Annu. Rev. Biomed. Eng., 2
(1), 715
–754
(2000). http://dx.doi.org/10.1146/annurev.bioeng.2.1.715 ARBEF7 1523-9829 Google Scholar
T. Hamaokaet al.,
“Near-infrared spectroscopy/imaging for monitoring muscle oxygenation and oxidative metabolism in healthy and diseased humans,”
J. Biomed. Opt., 12
(6), 062105
(2007). http://dx.doi.org/10.1117/1.2805437 JBOPFO 1083-3668 Google Scholar
M. WolfM. FerrariV. Quaresima,
“Progress of near-infrared spectroscopy and topography for brain and muscle clinical applications,”
J. Biomed. Opt., 12
(6), 062104
(2007). http://dx.doi.org/10.1117/1.2804899 JBOPFO 1083-3668 Google Scholar
M. FerrariL. MottolaV. Quaresima,
“Principles, techniques, and limitations of near infrared spectroscopy,”
Can. J. Appl. Physiol., 29
(4), 463
–487
(2004). http://dx.doi.org/10.1139/h04-031 CJAPEY 1066-7814 Google Scholar
R. Boushelet al.,
“Regional blood flow during exercise in humans measured by near-infrared spectroscopy and indocyanine green,”
J. Appl. Physiol., 89
(5), 1868
–1878
(2000). JAPYAA 0021-8987 Google Scholar
E. Rostrupet al.,
“Cerebral hemodynamics measured with simultaneous PET and near-infrared spectroscopy in humans,”
Brain Res., 954
(2), 183
–193
(2002). http://dx.doi.org/10.1016/S0006-8993(02)03246-8 BRREAP 1385-299X Google Scholar
L. C. Hensonet al.,
“Accuracy of a cerebral oximeter in healthy volunteers under conditions of isocapnic hypoxia,”
Anesthesiology, 88
(1), 58
–65
(1998). http://dx.doi.org/10.1097/00000542-199801000-00011 ANESAV 0003-3022 Google Scholar
Y. HoshiN. KobayashiM. Tamura,
“Interpretation of near-infrared spectroscopy signals: a study with a newly developed perfused rat brain model,”
J. Appl. Physiol., 90
(5), 1657
–1662
(2001). JAPYAA 0021-8987 Google Scholar
M. C. Van Beekveltet al.,
“Performance of near-infrared spectroscopy in measuring local O(2) consumption and blood flow in skeletal muscle,”
J. Appl. Physiol., 90
(2), 511
–519
(2001). JAPYAA 0021-8987 Google Scholar
J.-S. Wanget al.,
“Effects of normoxic and hypoxic exercise regimens on cardiac, muscular, and cerebral hemodynamics suppressed by severe hypoxia in humans,”
J. Appl. Physiol., 109
(1), 219
–229
(2010). http://dx.doi.org/10.1152/japplphysiol.00138.2010 JAPYAA 0021-8987 Google Scholar
A. Duncanet al.,
“Optical pathlength measurements on adult head, calf and forearm and the head of the newborn infant using phase resolved optical spectroscopy,”
Phys. Med. Biol., 40
(2), 295
–304
(1995). http://dx.doi.org/10.1088/0031-9155/40/2/007 PHMBA7 0031-9155 Google Scholar
K. MatsushitaS. HommaE. Okada,
“Influence of adipose tissue on muscle oxygenation measurement with NIRS instrument,”
Proc. SPIE, 3194 159
–165
(1998). http://dx.doi.org/10.1117/12.301048 SPIECJ 0361-0748 Google Scholar
H. H. Jasper,
“Report of the Committee on methods of clinical examination in electroencephalography. Appendix: the ten twenty electrode system of the international Federation,”
Electroencephalogr. Clin. Neurophysiol., 10 370
–375
(1957). ECNEAZ 0013-4694 Google Scholar
D. C. Krawczyk,
“Contributions of the prefrontal cortex to the neural basis of human decision making,”
Neurosci. Biobehav. Rev., 26
(6), 631
–664
(2002). http://dx.doi.org/10.1016/S0149-7634(02)00021-0 NBREDE 0149-7634 Google Scholar
E. K. MillerJ. D. Cohen,
“An integrative theory of prefrontal cortex function,”
Annu Rev. Neurosci., 24 167
–202
(2001). http://dx.doi.org/10.1146/annurev.neuro.24.1.167 ARNSD5 0147-006X Google Scholar
A. Duncanet al.,
“Measurement of cranial optical path length as a function of age using phase resolved near infrared spectroscopy,”
Pediatr. Res., 39
(5), 889
–894
(1996). http://dx.doi.org/10.1203/00006450-199605000-00025 PEREBL 0031-3998 Google Scholar
“Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology,”
Europ. Heart J., 17
(3), 354
–381
(1996). http://dx.doi.org/10.1093/oxfordjournals.eurheartj.a014868 EHJODF 0195-668X Google Scholar
M. Paganiet al.,
“Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog,”
Circ. Res., 59
(2), 178
–193
(1986). http://dx.doi.org/10.1161/01.RES.59.2.178 CIRUAL 0009-7330 Google Scholar
R. Roachet al.,
“The lake louise acute mountain sickness scoring system,”
Hypoxia and Molecular Medicine, 272
–274 Queen City Printers, Burlington, Vermont
(1993). Google Scholar
J. B. Sampsonet al.,
“Procedures for the measurement of acute mountain sickness,”
Aviat. Space Environ. Med., 54
(12 Pt 1), 1063
–1073
(1983). Google Scholar
S. Saitoet al.,
“Exercise-induced cerebral deoxygenation among untrained trekkers at moderate altitudes,”
Arch. Environ. Health, 54
(4), 271
–276
(1999). http://dx.doi.org/10.1080/00039899909602485 AEHLAU 0003-9896 Google Scholar
C. H. Imrayet al.,
“Near-infrared spectroscopy in the assessment of cerebral oxygenation at high altitude,”
Wilderness Environ. Med., 9
(4), 198
–203
(1998). http://dx.doi.org/10.1580/1080-6032(1998)009[0198:NISITA]2.3.CO;2 1080-6032 Google Scholar
T. RuppS. Perrey,
“Effect of severe hypoxia on prefrontal cortex and muscle oxygenation responses at rest and during exhaustive exercise,”
Adv. Exp. Med. Biol., 645 329
–334
(2009). http://dx.doi.org/10.1007/978-0-387-85998-9 AEMBAP 0065-2598 Google Scholar
A. W. SubudhiA. C. DimmenR. C. Roach,
“Effects of acute hypoxia on cerebral and muscle oxygenation during incremental exercise,”
J. Appl. Physiol., 103
(1), 177
–183
(2007). http://dx.doi.org/10.1152/japplphysiol.01460.2006 JAPYAA 0021-8987 Google Scholar
S. Y. Huanget al.,
“Internal carotid and vertebral arterial flow velocity in men at high altitude,”
J. Appl. Physiol., 63
(1), 395
–400
(1987). JAPYAA 0021-8987 Google Scholar
S. M. Otiset al.,
“Relationship of cerebral blood flow regulation to acute mountain sickness,”
J. Ultrasound Med., 8
(3), 143
–148
(1989). JUMEDA 0278-4297 Google Scholar
R. W. Baumgartneret al.,
“Enhanced cerebral blood flow in acute mountain sickness,”
Aviat. Space Environ. Med., 65
(8), 726
–729
(1994). Google Scholar
J. W. Severinghauset al.,
“Cerebral blood flow in man at high altitude. Role of cerebrospinal fluid pH in normalization of flow in chronic hypocapnia.,”
Circ. Res., 19
(2), 274
–282
(1966). http://dx.doi.org/10.1161/01.RES.19.2.274 CIRUAL 0009-7330 Google Scholar
Y. BhambhaniR. MalikS. Mookerjee,
“Cerebral oxygenation declines at exercise intensities above the respiratory compensation threshold,”
Respir. Physiol. Neurobiol., 156
(2), 196
–202
(2007). http://dx.doi.org/10.1016/j.resp.2006.08.009 RPNEAV 1569-9048 Google Scholar
K. Mølleret al.,
“Unchanged cerebral blood flow and oxidative metabolism after acclimatization to high altitude,”
J. Cereb. Blood Flow Metab., 22
(1), 118
–126
(2002). http://dx.doi.org/10.1097/00004647-200201000-00014 JCBMDN 0271-678X Google Scholar
T. Hayashiet al.,
“A theoretical model of oxygen delivery and metabolism for physiologic interpretation of quantitative cerebral blood flow and metabolic rate of oxygen,”
J. Cereb. Blood Flow Metab., 23
(11), 1314
–1323
(2003). http://dx.doi.org/10.1097/01.WCB.0000090506.76664.00 JCBMDN 0271-678X Google Scholar
R. Naeije,
“Physiological adaptation of the cardiovascular system to high altitude,”
Prog. Cardiovasc. Dis., 52
(6), 456
–466
(2010). http://dx.doi.org/10.1016/j.pcad.2010.03.004 PCVDAN 0033-0620 Google Scholar
I. HadoltG. Litscher,
“Noninvasive assessment of cerebral oxygenation during high altitude trekking in the Nepal Himalayas (2850–5600 m),”
Neurol. Res., 25
(2), 183
–188
(2003). http://dx.doi.org/10.1179/016164103101201175 NRESDZ 0161-6412 Google Scholar
A. W. Subudhiet al.,
“Frontal and motor cortex oxygenation during maximal exercise in normoxia and hypoxia,”
J. Appl. Physiol., 106
(4), 1153
–1158
(2009). http://dx.doi.org/10.1152/japplphysiol.91475.2008 JAPYAA 0021-8987 Google Scholar
J. E. Peltonenet al.,
“Cerebral and muscle tissue oxygenation in acute hypoxic ventilatory response test,”
Respir. Physiol. Neurobiol., 155
(1), 71
–81
(2007). http://dx.doi.org/10.1016/j.resp.2006.03.008 RPNEAV 1569-9048 Google Scholar
P. S. C. GomesC. MatsuuraY. N. Bhambhani,
“Effects of hypoxia on cerebral and muscle haemodynamics during knee extensions in healthy subjects,”
Europ. J. Appl. Physiol., 113
(1), 13
–23
(2012). http://dx.doi.org/10.1007/s00421-012-2408-7 EJAPFN 1439-6319 Google Scholar
I. Ellingsenet al.,
“Changes in human cerebral blood flow due to step changes in PAO2 and PACO2,”
Acta Physiol. Scand., 129
(2), 157
–163
(1987). http://dx.doi.org/10.1111/apha.1987.129.issue-2 APSCAX 0001-6772 Google Scholar
S. Bertugliaet al.,
“Hypoxia- or hyperoxia-induced changes in arteriolar vasomotion in skeletal muscle microcirculation,”
Am. J. Physiol., 260
(2 Pt 2), H362
–372
(1991). AJPHAP 0002-9513 Google Scholar
H. J. GrangerA. H. GoodmanB. H. Cook,
“Metabolic models of microcirculatory regulation,”
Fed. Proc., 34
(11), 2025
–2030
(1975). FEPRA7 0014-9446 Google Scholar
J. Vuyket al.,
“Acetazolamide improves cerebral oxygenation during exercise at high altitude,”
High Alt. Med. Biol., 7
(4), 290
–301
(2006). http://dx.doi.org/10.1089/ham.2006.7.290 HAMBB7 1527-0297 Google Scholar
P. Brassardet al.,
“Phenylephrine decreases frontal lobe oxygenation at rest but not during moderately intense exercise,”
J. Appl. Physiol., 108
(6), 1472
–1478
(2010). http://dx.doi.org/10.1152/japplphysiol.01206.2009 JAPYAA 0021-8987 Google Scholar
J. A. VogelC. W. Harris,
“Cardiopulmonary responses of resting man during early exposure to high altitude,”
J. Appl. Physiol., 22
(6), 1124
–1128
(1967). JAPYAA 0021-8987 Google Scholar
M. Buchheitet al.,
“Effect of acute hypoxia on heart rate variability at rest and during exercise,”
Int. J. Sports Med., 25
(4), 264
–269
(2004). http://dx.doi.org/10.1055/s-2004-819938 IJSMDA 0172-4622 Google Scholar
M. Willeet al.,
“Changes in cardiac autonomic activity during a passive 8 hour acute exposure to 5500 m normobaric hypoxia are not related to the development of acute mountain sickness,”
Int. J. Sports Med., 33
(3), 186
–191
(2012). http://dx.doi.org/10.1055/s-0031-1291325 IJSMDA 0172-4622 Google Scholar
Y.-C. Chenet al.,
“Effect of rapid ascent to high altitude on autonomic cardiovascular modulation,”
Am. J. Med. Sci., 336
(3), 248
–253
(2008). http://dx.doi.org/10.1097/MAJ.0b013e3181629a32 AJMEAZ 0002-9343 Google Scholar
M. BurtscherE. BrandstätterH. Gatterer,
“Preacclimatization in simulated altitudes,”
Sleep Breath., 12
(2), 109
–114
(2008). http://dx.doi.org/10.1007/s11325-007-0127-9 1520-9512 Google Scholar
J. H. Klaessenset al.,
“Effects of skin on bias and reproducibility of near-infrared spectroscopy measurement of cerebral oxygenation changes in porcine brain,”
J. Biomed. Opt., 10
(4), 044003
(2005). http://dx.doi.org/10.1117/1.1989315 JBOPFO 1083-3668 Google Scholar
H. ObrigA. Villringer,
“Beyond the visible—imaging the human brain with light,”
J. Cereb. Blood Flow Metab., 23
(1), 1
–18
(2003). http://dx.doi.org/10.1097/00004647-200301000-00001 JCBMDN 0271-678X Google Scholar
D. BauerR. GrebeA. Ehrlacher,
“A new method to model change in cutaneous blood flow due to mechanical skin irritation part I: comparison between experimental and numerical data,”
J. Theor. Biol., 238
(3), 575
–587
(2006). http://dx.doi.org/10.1016/j.jtbi.2005.06.023 JTBIAP 0022-5193 Google Scholar
T. Takahashiet al.,
“Influence of skin blood flow on near-infrared spectroscopy signals measured on the forehead during a verbal fluency task,”
NeuroImage, 57
(3), 991
–1002
(2011). http://dx.doi.org/10.1016/j.neuroimage.2011.05.012 NEIMEF 1053-8119 Google Scholar
D. A. BoasA. M. DaleM. A. Franceschini,
“Diffuse optical imaging of brain activation: approaches to optimizing image sensitivity, resolution, and accuracy,”
NeuroImage, 23
(Suppl 1), S275
–288
(2004). http://dx.doi.org/10.1016/j.neuroimage.2004.07.011 NEIMEF 1053-8119 Google Scholar
T. AckerH. Acker,
“Cellular oxygen sensing need in CNS function: physiological and pathological implications,”
J. Exp. Biol., 207
(18), 3171
–3188
(2004). http://dx.doi.org/10.1242/jeb.01075 JEBIAM 0022-0949 Google Scholar
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||

