|
|
|
Multiphoton microscopy (MPM) has become an indispensable optical imaging modality due to its intrinsic three-dimensional localization, low photobleaching and photodamage outside the focal volume, and improved imaging depth.1 MPM typically uses near-infrared (NIR) excitation at to generate fluorescence in the visible wavelengths.2 Although the excitation wavelength of MPM has been extended3, 4, 5, 6, 7, 8 beyond , to our knowledge no imaging has been performed with NIR-emitting contrast agents. Conventional (linear) fluorescence imaging has shown that molecular imaging with NIR agents minimizes the contribution from endogenous fluorescence and increases penetration depth.9 Since conventional MPM efficiently images endogenous molecules such as collagen, elastin, and reduced nicotinamide adenine dinucleotide (NADH),10 the signal from these and other endogenous fluorophores restricts molecular imaging with exogenous contrast agents. This is particularly problematic when target concentrations are low, and strategies to reduce autofluorescence become critical to image contrast. Here, we measure two-photon-induced fluorescence of NIR dyes and demonstrate their use for autofluorescence-free biological imaging. Heptamethine cyanine dyes were selected as NIR contrast agents because of their biocompatibility and the availability of diverse structures with single photon (1P) absorption and emission between 700 and . Cypate, a derivative of indocyanine green, was prepared as previously reported,11 and -diethylthiatricarbocyanine iodide (DTTCI) were purchased from a commercial source (Sigma-Aldrich, Saint Louis, Missouri). These dyes exhibit an absorption/emission peak of and , respectively. Fortuitously, these absorption maxima of NIR dyes correspond to roughly half the telecommunications spectral window at . As this overlaps with the gain spectrum of erbium, we perform all-NIR MPM using a simple, turnkey erbium-doped fiber laser. A mode-locked femtosecond fiber laser (Mercury 1000, PolarOnyx, Sunnyvale, California ) provided of excitation light at through a optical fiber (Fig. 1 ). At the output of the fiber, the pulse duration was nominally with a repetition rate of , although positive material dispersion of the optical components in the system leads to pulse broadening and reduces excitation efficiency at the sample. These losses can be recovered if the pulses arriving at the sample are transform-limited by precompensating for material dispersion in the system,12 for example with a prism pair. In the present scheme, since the laser is designed to provide transform-limited pulses at the output of a fiber, we reduced the length of the fiber (to ) as a simple, albeit not continuously variable, method of dispersion precompensation. Fig. 1Schematic diagram of all-NIR multiphoton microscope. PD, photodiode; INV, inverter; ND, neutral density filter; DM, dichroic mirror; OBJ, objective lens; RG9, Schott glass filter; PMT, photomultiplier tube; and TCSPC, time-correlated single photon counting card. 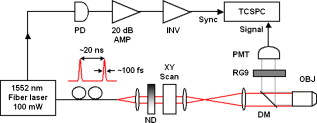 The laser light was collimated and passed through neutral density filters for controlling the incident power on the sample (between ). The beam was then expanded , directed through a dichroic mirror (LP02-980RS, Semrock, Rochester, New York ), and focused onto the sample using a , Plan-Apochromat (Zeiss) objective lens. Two-photon (2P) epifluorescence from the sample was collected through the same objective and directed toward the detector using the dichroic mirror. The detector was a thermoelectrically cooled, red-enhanced photomultiplier tube (PMT) (PMC-100-20, Becker-Hickl, Berlin, Germany ). The seed monitor of the laser triggered data acquisition using a time-correlated single photon counting (TCSPC) card (SPC-730, Becker-Hickl). Although the PMT cathode sensitivity appeared to be negligible at the excitation wavelength, residual excitation light was further removed using a -thick RG9 Schott glass filter. This filter also served to minimize stray light in the visible wavelength range. Images were acquired by raster scanning the excitation beam with a galvanometric mirror pair (GSI Group, Bedford, Massachusetts), centered near a conjugate plane of the back focal plane of the objective lens to approximate telecentric scanning and thus a flat field of view (FOV). The pixel integration time varied from , limited by TCSPC. Collected images were analyzed with a single photon counting module (SPCM) and SPCImage software (Becker-Hickl). The total fluorescence photon count scaled quadratically with the average incident power at the sample [a slope of on a log-log plot, Fig. 2 ], verifying that the measured signals were indeed due to a 2P process.1 Using fluorescently labeled particles embedded in agarose gel, the lateral and axial resolutions [Figs. 2 and 2] were measured as and , respectively, which agreed to within 5% of the theoretical calculation.13 The longer emission wavelengths in this system result in to lower resolution than MPM of visible dyes. Fig. 2(a) Quadratic dependence of fluorescence on excitation power indicates 2P process. Error bars in the abscissa are , determined by the power meter resolution. Error bars in the ordinate are the standard deviation of three measurements. (b) Lateral and (c) axial point spread functions (PSFs). 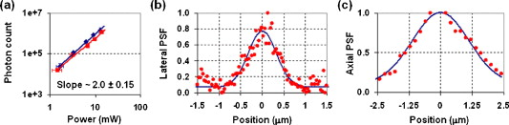 Having thus attributed NIR fluorescence to 2P excitation, we explored the feasibility of biological imaging with these dyes. All animal studies were performed in compliance with the Washington University School of Medicine Animal Studies Committee requirements for the humane care and use of laboratory animals in research. Human epithelial carcinoma xenografts were grown in nude mice by injection of A431 cells. Tumor tissue was harvested and snap-frozen. cryostat sections were stained with cypate or DTTCI by incubating with of dye in phosphate buffered saline (PBS) for , and rinsed with PBS three times for each. Coverslips were applied and sealed with nail polish. Figure 3 presents all-NIR 2P images ) of -thick sections of human epithelial carcinoma tissues labeled with the NIR dyes, which localized in the cell membranes and stromal tissues but did not stain the nuclei, shown as dark regions. The benefits of all-NIR imaging are observed by imaging unlabelled samples (Fig. 4 ) using a wide-field upright 1P microscope (Zeiss Axio Imager.Z1, Plan-Apochromat objective) at for NIR excitation and for visible excitation. A comparison of histograms and images, using the same intensity color scale, indicated a dramatic reduction in NIR autofluorescence. In practice, reducing the background should result in higher contrast imaging, in particular for targeted molecular imaging. Since water absorption is higher at than at conventional MPM excitation wavelengths, the possibility of thermal damage both within and outside the focus needs to be explored. In preliminary imaging with ten samples, no heating effects were observed from repeated imaging. Further studies are needed to examine the potentially deleterious effects of imaging in this spectral band, although previous reports at this excitation wavelength4, 5 have likewise not reported damage at low power levels. Another potential disadvantage of imaging in this spectral window is the lower quantum efficiency and higher dark noise of detectors in the NIR, which may result in lower contrast imaging. Fig. 3All-NIR 2P fluorescence microscopy images of A431 human carcinoma tissue sections labeled with NIR dyes (a) and (b) DTTCI and (c) and (d) cypate. Incident power was . The scale bar is . (Color online only.) 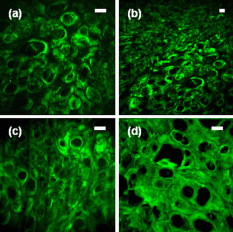 Fig. 4Dramatically attenuated NIR autofluorescence is exhibited by unlabeled A431 human epithelial cells under NIR and visible 1P excitation. Intensities are shown on the same color scale for comparison. 2P imaging of unlabeled samples similarly yielded blank images. Field of view . (Color online only.) 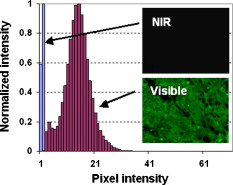 We have developed an all-NIR MPM instrument for imaging dyes with emission above . Imaging of NIR fluorophores expands the available spectrum for MPM, minimizes autofluorescence, and may potentially improve penetration depth. This simple and affordable turnkey fiber-based laser system could serve as a platform for characterizing and imaging other NIR fluorophores14, 15 near . While 2P excitation spectra do not consistently mirror the 1P absorption spectra,2 in many cases the 2P excitation peak is at or near twice the wavelength of the 1P peak. Therefore, dyes with 1P peaks near , or half the 2P excitation laser in our experiment, are generally expected to demonstrate a high 2P action cross section. The use of tunable lasers would provide flexibility in the choice of NIR fluorescent dyes for all-NIR MPM, improve 2P cross sections, and enhance the resultant fluorescence by exciting the molecules at their maximum 2P absorption wavelengths. In addition, spectroscopic detection using an optical parametric oscillator4 can be used to identify the optimal excitation wavelength of the dyes. Alternatively, dyes can be engineered for improved 2P absorption within the NIR spectral range of our current instrument. NIR emission does not compromise many existing advantages of 2P microscopy, such as multiplexing of fluorophores. Multiplexed detection can be achieved by adapting the system to cover additional spectral ranges, for example, by combining the NIR light source with conventional Ti:sapphire lasers or novel fiber-based or solid state light sources.16, 17, 18 Frequency doubling of the fiber laser would result in a second excitation wavelength at , comparable to conventional MPM, in addition to the fundamental wavelength near . The former can be used to excite visible dyes, and the latter NIR dyes. Multimodal microscopy is feasible on this imaging platform, combining 2P with higher order MPM of visible dyes,4 harmonic generation,3 optical coherence microscopy,10 or autoconfocal microscopy.19 In conclusion, we have demonstrated 2P microscopy with NIR contrast agents and a fiber laser at . This system widens the spectral window available for molecular imaging with MPM and virtually eliminates autofluorescence. We anticipate that this technology can be rapidly adopted for use in intravital imaging of living tissues that are accessible to endoscopes and catheters. AcknowledgmentsThis study was supported by a grant from the National Institutes of Health (R21/R33 CA12353701). We acknowledge S. Bhaumik, A. Sood, and J. DePuy for help in sample preparation, and P. Codella for performing linear spectroscopy. ReferencesW. Denk, J. H. Strickler, and W. W. Webb,
“Two-photon laser scanning fluorescence microscopy,”
Science, 248 73
–76
(1990). https://doi.org/10.1126/science.2321027 0036-8075 Google Scholar
C. Xu and W. W. Webb,
“Measurement of two-photon excitation cross sections of molecular fluorophores with data from ,”
J. Opt. Soc. Am. B, 13 481
–491
(1996). https://doi.org/10.1364/JOSAB.13.000481 0740-3224 Google Scholar
S. W. Chu, I. H. Chen, T. M. Liu, P. C. Chen, C. K. Sun, and B. L. Lin,
“Multimodal nonlinear spectral microscopy based on a femtosecond Cr:forsterite laser,”
Opt. Lett., 26 1909
–1911
(2001). https://doi.org/10.1364/OL.26.001909 0146-9592 Google Scholar
G. McConnell,
“Nonlinear optical microscopy at wavelengths exceeding using a synchronously pumped femtosecond-pulsed optical parametric oscillator,”
Phys. Med. Biol., 52 717
–724
(2007). https://doi.org/10.1088/0031-9155/52/3/013 0031-9155 Google Scholar
D. Träutlein, F. Adler, K. Moutzouris, A. Jeromin, A. Leitenstorfer, and E. Ferrando-May,
“Highly versatile confocal microscopy system based on a tunable femtosecond Er:fiber source,”
J. Biophoton., 1 53
–61
(2008). https://doi.org/10.1002/jbio.200710019 Google Scholar
M. Balu, T. Baldacchini, J. Carter, T. B. Krasieva, R. Zadoyan, and B. J. Tromberg,
“Effect of excitation wavelength on penetration depth in nonlinear optical microscopy of turbid media,”
J. Biomed. Opt., 14 010508
(2009). https://doi.org/10.1117/1.3081544 1083-3668 Google Scholar
V. Andresen, S. Alexander, W. H. Heupel, M. Hirschberg, R. M. Hoffman, and P. Friedl,
“Infrared multiphoton microscopy: subcellular-resolved deep tissue imaging,”
Curr. Opin. Biotechnol., 20 54
–62
(2009). https://doi.org/10.1016/j.copbio.2009.02.008 0958-1669 Google Scholar
D. Kobat, M. E. Durst, N. Nishimura, A. W. Wong, C. B. Schaffer, and C. Xu,
“Deep tissue multiphoton microscopy using longer wavelength excitation,”
Opt. Express, 17 13354
–13364
(2009). https://doi.org/10.1364/OE.17.013354 1094-4087 Google Scholar
E. M. Sevick-Muraca and J. C. Rasmussen,
“Molecular imaging with optics: primer and case for near-infrared fluorescence techniques in personalized medicine,”
J. Biomed. Opt., 13 041303
(2008). https://doi.org/10.1117/1.2953185 1083-3668 Google Scholar
S. Yazdanfar, Y. Y. Chen, P. T. C. So, and L. H. Laiho,
“Multifunctional imaging of endogenous contrast by simultaneous nonlinear and optical coherence microscopy of thick tissues,”
Microsc. Res. Tech., 70 628
–633
(2007). https://doi.org/10.1002/jemt.20447 1059-910X Google Scholar
S. Achilefu, R. B. Dorshow, J. E. Bugaj, and R. Rajagopalan,
“Novel receptor-targeted fluorescent contrast agents for in vivo tumor imaging,”
Invest. Radiol., 35 479
–485
(2000). https://doi.org/10.1097/00004424-200008000-00004 0020-9996 Google Scholar
D. K. Kim, H. J. Choi, S. Yazdanfar, and P. T. C. So,
“Ultrafast optical pulse delivery with fibers for nonlinear microscopy,”
Microsc. Res. Tech., 71 887
–896
(2008). https://doi.org/10.1002/jemt.20634 1059-910X Google Scholar
M. Gu,
“Image formation in multiphoton fluorescence microscopy,”
Handbook of Biomedical Nonlinear Optical Microscopy, 266
–282 Oxford University Press, New York
(2008). Google Scholar
Y. P. Ye, S. Bloch, B. G. Xu, and S. Achilefu,
“Design, synthesis, and evaluation of near infrared fluorescent multimeric RGD peptides for targeting tumors,”
J. Med. Chem., 49 2268
–2275
(2006). https://doi.org/10.1021/jm050947h 0022-2623 Google Scholar
M. Y. Berezin, H. Lee, W. Akers, and S. Achilefu,
“Near infrared dyes as lifetime solvatochromic probes for micropolarity measurements of biological systems,”
Biophys. J., 93 2892
–2899
(2007). https://doi.org/10.1529/biophysj.107.111609 0006-3495 Google Scholar
J. R. Unruh, E. S. Price, R. G. Molla, L. Stehno-Bittel, C. K. Johnson, and R. Q. Hui,
“Two-photon microscopy with wavelength switchable fiber laser excitation,”
Opt. Express, 14 9825
–9831
(2006). https://doi.org/10.1364/OE.14.009825 1094-4087 Google Scholar
S. Sakadžic, U. Demirbas, T. R. Mempel, A. Moore, S. Ruvinskaya, D. A. Boas, A. Sennaroglu, F. X. Kartner, and J. G. Fujimoto,
“Multi-photon microscopy with a low-cost and highly efficient Cr:LiCAF laser,”
Opt. Express, 16 20848
–20863
(2008). https://doi.org/10.1364/OE.16.020848 1094-4087 Google Scholar
S. Tang, J. Liu, T. B. Krasieva, Z. P. Chen, and B. J. Tromberg,
“Developing compact multiphoton systems using femtosecond fiber lasers,”
J. Biomed. Opt., 14 030508
(2009). https://doi.org/10.1117/1.3153842 1083-3668 Google Scholar
C. Joo, C. Zhan, Q. Li, and S. Yazdanfar,
“Autoconfocal transmission microscopy based on two-photon induced photocurrent of Si photodiodes,”
Opt. Lett., 35 67
–69
(2010). https://doi.org/10.1364/OL.35.000067 0146-9592 Google Scholar
|


